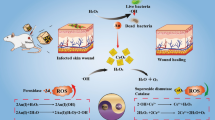
Abstract
Rapid, convenient, and sensitive detection of bacteria and development of novel antibacterial materials are conducive to accurate treatment of bacterial infection and reducing the generation of drug-resistant bacteria caused by overuse of antibiotics. A dual-function magnetic nanozyme, Fc-MBL@rGO@Fe3O4, has been constructed with broad-spectrum bacterial affinity and good peroxidase-like activity. Detection signal amplification was realized in the presence of 3,3’,5,5’-tetramethylbenzidine (TMB) with a detection limit of 26 CFU/mL. In addition, the excellent photothermal properties of Fc-MBL@rGO@Fe3O4 could realize synergistic chemodynamic/photothermal antibacterial therapy. Furthermore, the good bacterial affinity of Fc-MBL@rGO@Fe3O4 enhances the accurate and rapid attack of hydroxyl radical (·OH) on the bacterial membrane and achieves efficient sterilization (100%) at low concentration (40 µg/mL) and mild temperature (47℃). Notably, Fc-MBL@rGO@Fe3O4 has a broad spectrum of antibacterial activity against Gram-negative, Gram-positive, and drug-resistant bacteria. The magnetic nanoplatform integrating detection-sterilization not only meets the need for highly sensitive and accurate detection in different scenarios, but can realize low power density NIR-II light-responsive chemodynamic/photothermal antibacterial therapy, which has broad application prospects.
Graphical abstract







Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Andersson DI, Hughes D (2010) Antibiotic resistance and its cost: is it possible to reverse resistance? Nat Rev Microbiol 8(4):260–271
Tang WH, Ranganathan N, Shahrezaei V, Larrouy-Maumus G (2019) MALDI-TOF mass spectrometry on intact bacteria combined with a refined analysis framework allows accurate classification of MSSA and MRSA. PLoS ONE 14(6):e0218951
Kerremans JJ, Verboom P, Stijnen T, Roijen LHV, Goessens W, Verbrugh HA, Vos MC (2008) Rapid identification and antimicrobial susceptibility testing reduce antibiotic use and accelerate pathogen-directed antibiotic use. J Antimicrob Chemoth 61(2):428–435
Altun O, Botero-Kleiven S, Carlsson S, Ullberg M, Ozenci V (2015) Rapid identification of bacteria from positive blood culture bottles by MALDI-TOF MS following short-term incubation on solid media. J Med Microbiol 64:1346–1352
Loesche WJ, Lopatin DE, Stoll J, Vanpoperin N, Hujoel PP (1992) Comparison of various detection methods for periodontopathic bacteria - can culture be considered the primary reference-standard. J Clin Microbiol 30(2):418–426
Zhang H, Zhang YL, Lin YK, Liang TW, Chen ZH, Li JF, Yue ZF, Lv JZ, Jiang Q, Yi CQ (2015) Ultrasensitive detection and rapid identification of multiple foodborne pathogens with the naked eyes. Biosens Bioelectron 71:186–193
Wang YL, Li QY, Zhang R, Tang KQ, Ding CF, Yu SN (2020) SERS-based immunocapture and detection of pathogenic bacteria using a boronic acid-functionalized polydopamine-coated Au@Ag nanoprobe. Microchim Acta 187(5):209
Shi HM, Sun JJ, Han RR, Ding CF, Hu FP, Yu SN (2020) The strategy for correcting interference from water in Fourier transform infrared spectrum based bacterial typing. Talanta 208:120347
Muhamadali H, Subaihi A, Mohammadtaheri M, Xu Y, Ellis DI, Ramanathan R, Bansal V, Goodacre R (2016) Rapid, accurate, and comparative differentiation of clinically and industrially relevant microorganisms via multiple vibrational spectroscopic fingerprinting. Analyst 141(17):5127–5136
Das R, Dhiman A, Kapil A, Bansal V, Sharma TK (2019) Aptamer-mediated colorimetric and electrochemical detection of Pseudomonas aeruginosa utilizing peroxidase-mimic activity of gold Nanozyme. Anal Bioanal Chem 411(6):1229–1238
Li X, Wang LJ, Du D, Ni L, Pan JM, Niu XH (2019) Emerging applications of nanozymes in environmental analysis: opportunities and trends. Trac-Trend Anal Chem 120:115653
Azevedo AM, Martins VC, Prazeres DM, Vojinovic V, Cabral JM, Fonseca LP (2003) Horseradish peroxidase: a valuable tool in biotechnology. Biotechnol Annu Rev 9:199–247
van Beilen JB, Li Z (2002) Enzyme technology: an overview. Curr Opin Biotech 13(4):338–344
Chen ZW, Wang ZZ, Ren JS, Qu XG (2018) Enzyme mimicry for combating bacteria and biofilms. Acc Chem Res 51(3):789–799
Lyu Z, Zhou J, Ding S, Du D, Wang J, Liu Y, Lin Y (2023) Recent advances in single-atom nanozymes for colorimetric biosensing. Trac-Trend Anal Chem 168:117280
Kuah E, Toh S, Yee J, Ma Q, Gao ZQ (2016) Enzyme mimics: advances and applications. Chem-Eur J 22(25):8404–8430
Li X, Zhu HJ, Liu P, Wang MZ, Pan JM, Qiu FX, Ni L, Ni XH (2021) Realizing selective detection with nanozymes: strategies and trends. Trac-Trend Anal Chem 143:116379
Feng YY, Qin J, Zhou Y, Yue Q, Wei J (2022) Spherical mesoporous Fe-N-C single-atom nanozyme for photothermal and catalytic synergistic antibacterial therapy. J Colloid Interf Sci 606:826–836
Yin W, Yu J, Lv F, Yan L, Zheng LR, Gu Z, Zhao YJAN (2016) Functionalized nano-MoS2 with peroxidase catalytic and near-infrared photothermal activities for safe and synergetic wound antibacterial applications. ACS Nano 10:11000–11011
Wang X, Sun XY, Bu T, Wang QZ, Zhang H, Jia P, Li LW, Wang L (2021) Construction of a photothermal hydrogel platform with two-dimensional PEG@zirconium-ferrocene MOF nanozymes for rapid tissue repair of bacteria-infected wounds. Acta Biomater 135:342–355
Zhu YW, Xu C, Zhang N, Ding XK, Yu BR, Xu FJ (2018) Polycationic synergistic antibacterial agents with multiple functional components for efficient anti-infective therapy. Adv Funct Mater 28(14):1706709
Yang Y, Deng YY, Huang JB, Fan X, Cheng C, Nie CX, Ma L, Zhao WF, Zhao CS (2019) Size-transformable Metal-Organic Framework-derived nanocarbons for localized chemo-photothermal bacterial ablation and wound disinfection. Adv Funct Mater 29(33):1900143
Jeong CJ, Sharker SM, In I, Park SY (2015) Iron oxide@PEDOT-based recyclable photothermal nanoparticles with poly(vinylpyrrolidone) sulfobetaines for rapid and effective antibacterial activity. ACS Appl Mater Interfaces 7(18):9469–9478
Chen LF, Xing SH, Lei YL, Chen QS, Zou Z, Quan K, Qing ZH, Liu JW, Yang RH (2021) A glucose-powered activatable nanozyme breaking pH and H2O2 limitations for treating diabetic infections. Angew Chem Int Ed 60(44):23534–23539
Deng JQ, Xu JY, Ouyang MZ, Zou Z, Lei YL, Li JB, Qing ZH, Yang RH (2022) Target-triggered hairpin-free chain-branching growth of DNA dendrimers for contrast-enhanced imaging in living cells by avoiding signal dispersion. Chin Chem Lett 33:773–777
Yingnan Z, Yaqing X, Zhang, Yalan, Tong, Taotao W (2019) Multifunctional magnetic copper ferrite nanoparticles as fenton-like reaction and near-infrared photothermal agents for synergetic antibacterial therapy. ACS Appl Mater Interfaces 11:31649–31660
Wu HH, Cheng KM, He Y, Li ZY, Su HL, Zhang XM, Sun YA, Shi W, Ge DT (2019) Fe3O4-based multifunctional nanospheres for amplified magnetic targeting photothermal therapy and fenton reaction. Acs Biomater Sci Eng 5(2):1045–1056
Gao Q, Zhang X, Yin WY, Ma DQ, Xie CJ, Zheng LR, Dong XH, Mei LQ, Yu J, Wang CZ, Gu ZJ, Zhao YL (2018) Functionalized MoS2 nanovehicle with near-infrared laser-mediated nitric oxide release and photothermal activities for advanced bacteria-infected wound therapy. Small 14(45):1802290
Gao LZ, Zhuang J, Nie L, Zhang JB, Zhang Y, Gu N, Wang TH, Feng J, Yang DL, Perrett S, Yan X (2007) Intrinsic peroxidase-like activity of ferromagnetic nanoparticles. Nat Nanotechnol 2(9):577–583
Lutz JF, Stiller S, Hoth A, Kaufner L, Pison U, Cartier R (2006) One-pot synthesis of PEGylated ultrasmall iron-oxide nanoparticles and their in vivo evaluation as magnetic resonance imaging contrast agents. Biomacromolecules 7(11):3132–3138
Morales-Narvaez E, Merkoci A (2019) Graphene oxide as an optical biosensing platform: a progress report. Adv Mater 31(6):1805043
Ghosal K, Sarkar K (2018) Biomedical applications of graphene nanomaterials and beyond. ACS Biomater Sci Eng 4(8):2653–2703
Neth O, Jack DL, Dodds AW, Holzel H, Klein NJ, Turner MW (2000) Mannose-binding lectin binds to a range of clinically relevant microorganisms and promotes complement deposition. Infect Immun 68(2):688–693
Xue YH, Chen H, Yu DS, Wang SY, Yardeni M, Dai QB, Guo MM, Liu Y, Lu F, Qu J, Dai LM (2011) Oxidizing metal ions with graphene oxide: the in situ formation of magnetic nanoparticles on self-reduced graphene sheets for multifunctional applications. Chem Commun 47(42):11689–11691
Yu MH, Zhang L, Qian GQ, Shi HM, Yu SN (2022) Fc-MBL-modified Fe3O4 magnetic bead enrichment and fixation in Gram strain for rapid detection of low-concentration bacteria. Microchim Acta 189:169
Funding
This work was supported by the National Key Research and Development Plan of China (2018YFF0212501) and National Natural Science Foundation of China (31470786).
Author information
Authors and Affiliations
Contributions
Yuyan Xue: Conceptualization, Methodology, Data curation, Writing - original draft preparation. Qiaoyu Li: Software, Formal analysis. Yanlin Wang: Formal analysis. Hao Shen: Visualization, Reviewing, Validation, Supervision. Shaoning Yu: Editing, Supervision.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xue, Y., Li, Q., Wang, Y. et al. A magnetic nanozyme platform for bacterial colorimetric detection and chemodynamic/photothermal synergistic antibacterial therapy. Microchim Acta 191, 214 (2024). https://doi.org/10.1007/s00604-024-06270-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-024-06270-y