Abstract
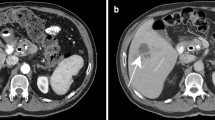
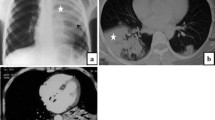
The treatment of fungal infections presents significant challenges due to the lack of standardized diagnostic procedures, a restricted range of antifungal treatments, and the risk of harmful interactions between antifungal medications and the immunosuppressive drugs used in anti-inflammatory treatment for critically ill patients with COVID-19. Mucormycosis and aspergillosis are the primary invasive fungal infections in patients with severe COVID-19, occurring singly or in combination. Histopathological examination is a vital diagnostic technique that details the presence and invasion of fungi within tissues and blood vessels, and the body’s response to the infection. However, the pathology report omits information on the most common fungi associated with the observed morphology, as well as other potential fungi and parasites that ought to be included in the differential diagnosis. This research marks significance in diagnosing fungal infections, such as mucormycosis and aspergillosis associated to COVID-19 by field emission scanning electron microscopy (FESEM) imaging to examine unstained histopathology slides, allowing for a detailed morphological analysis of the fungus. FESEM provides an unprecedented resolution and detail, surpassing traditional Hematoxylin & Eosin (H&E) and Grocott’s Methenamine Silver (GMS) staining methods in identifying and differentiating dual fungal infections and diverse fungal species. The findings underscore the critical need for individualized treatment plans for patients severely affected by COVID-19 and compounded by secondary fungal infections. The high-magnification micrographs reveal specific fungal morphology and reproductive patterns. Current treatment protocols largely depend on broad-spectrum antifungal therapies. However this FESEM guided diagnostic approach can help in targeted patient specific anti fungal therapies. Such precision could lead to more effective early interventions, addressing the complex management required for severe COVID-19 cases with coexisting fungal infections. This approach significantly advances disease management and patient recovery, advocating for personalized, precision medicine in tackling this multifaceted clinical challenge.
Graphical Abstract






Similar content being viewed by others
Data Availability
All data that support the findings of this study are included within the article.
Abbreviations
- ACE2:
-
Angiotensin converting enzyme 2
- CAA:
-
COVID-19 Associated Aspergillosis
- CAM:
-
COVID-19 Associated Mucormycosis
- CCR2+:
-
C-C chemokine receptor type 2
- COVID-19:
-
CoronaVirus Disease of 2019
- FEG:
-
Field Emission Gun
- FESEM:
-
Field Emission Scanning Electron Microscope
- GAG:
-
Galactosaminogalactan
- GM:
-
Galactomannan
- GMS Stain:
-
Grocott-Gömöri'sMethenamine Silver stain
- H&E Stain:
-
Hematoxylin and eosin stain
- HIF-1α:
-
Hypoxia-Inducible Factor 1α
- HMDS:
-
Hexamethyldisilane
- ICU:
-
Intensive care unit
- IFN:
-
Interferon
- IFN-γ:
-
Interferon-gamma
- IL-1ß:
-
Interleukin 1-beta
- IL-6:
-
Interleukin-6
- LCB:
-
Lactophenol cotton blue
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate
- NLRs:
-
NOD like receptors
- PAS:
-
Periodic acid schiff
- PCR:
-
Polymerase chain reaction
- PRRs:
-
Pattern recognition receptors
- SARS-CoV-2:
-
Severe Acute Respiratory Syndrome Coronavirus 2
- TNF-α:
-
Tumor Necrosis Factor-alpha
- TLR4:
-
Toll-like receptor 4
- WBC:
-
White blood cells
References
Islam MR, Rahman MM, Ahasan MT et al (2022) The impact of mucormycosis (black fungus) on SARS-CoV-2-infected patients: at a glance. Environ Sci Pollut Res 29:69341–69366. https://doi.org/10.1007/s11356-022-22204-8
Ganesan N, Sivanandam S (2022) Histomorphological features of mucormycosis with rise and fall of COVID-19 pandemic. Pathol Res Pract 236:153981. https://doi.org/10.1016/j.prp.2022.153981
Akhtar N, Wani AK, Tripathi SK, Prakash A, Mannan MA (2022) The role of SARS-CoV-2 immunosuppression and the therapy used to manage COVID-19 disease in the emergence of opportunistic fungal infections: a review. Current Res Biotechnol. https://doi.org/10.1016/j.crbiot.2022.08.001
Habibzadeh A, Lankarani KB, Farjam M et al (2022) Prevalence of fungal drug resistance in COVID-19 Infection: a global meta-analysis. Curr Fungal Infect Rep 16:154–164. https://doi.org/10.1007/s12281-022-00439-9
Qin C, Zhou L, Hu Z, Zhang S, Yang S, Tao Y, Xie C, Ma K, Shang K, Wang W, Tian DS (2020) Dysregulation of immune response in patients with coronavirus 2019 (COVID-19) in Wuhan. China Clin Infect Dis 71:762–768. https://doi.org/10.1093/cid/ciaa248
Hall MW et al (2022) Immune immunomodulation in coronavirus disease 2019 (COVID-19): strategic considerations for personalized therapeutic intervention. Clin Infect Dis 74:144–148. https://doi.org/10.1093/cid/ciaa904
Jin Y, Ji W, Yang H et al (2020) Endothelial activation and dysfunction in COVID-19: from basic mechanisms to potential therapeutic approaches. Sig Transduct Target Ther 5:293. https://doi.org/10.1038/s41392-020-00454-7
Hoenigl M, Seidel D, Carvalho A, Rudramurthy SM, Arastehfar A, Gangneux JP, Nasir N, Bonifaz A, Araiza J, Klimko N, Serris A (2022) The emergence of COVID-19 associated mucormycosis: a review of cases from 18 countries. Lancet Microbe. https://doi.org/10.1016/S2666-5247(21)00237-8
Baral PK, Aziz MA, Islam MS (2022) Comparative risk assessment of COVID-19 associated mucormycosis and aspergillosis: a systematic review. Health Sci Rep 5:e789. https://doi.org/10.1002/hsr2.789
Sundeep D, Varadharaj EK, Umadevi K, Jhansi R (2023) Role of nanomaterials in screenprinted electrochemical biosensors for detection of COVID-19 and for post-covid syndromes. ECS Adv 2:016502. https://doi.org/10.1149/2754-2734/acb832
Lass-Flörl C (2009) Zygomycosis: conventional laboratory diagnosis. Clin Microbiol Infect 15:60–65. https://doi.org/10.1111/j.1469-0691.2009.02999.x
Moran CA, Mullick FG, Ishak KG, Johnson FB, Hummer WB (1991) Identification of titanium in human tissues: probable role in pathologic processes. Hum Pathol 22:450–454. https://doi.org/10.1016/0046-8177(91)90130-H
Iwen PC, Sigler L, Noel RK, Freifeld AG (2007) Mucorcircinelloides was identified by molecular methods as a cause of primary cutaneous zygomycosis. J Clin Microbiol 45:636–640. https://doi.org/10.1128/jcm.01907-06
Umadevi K, Sundeep D, Jhansi R et al (2023) Effect of functionalization of 2D graphene nanosheets on oxidation stress of BEAS-2B cells. BioNanoSci. https://doi.org/10.1007/s12668-023-01155-5
Ibrahim AS, Spellberg B, Walsh TJ, Kontoyiannis DP (2012) Pathogenesis of mucormycosis. Clin Infect Dis 54:S16–S22. https://doi.org/10.1093/cid/cir865
Skiada A, Pavleas I, Drogari-Apiranthitou M (2020) Epidemiology and diagnosis of mucormycosis: an update. J Fungi (Basel) 6:265. https://doi.org/10.3390/jof6040265
Reiterer M, Rajan M, Gómez-Banoy N, Lau JD, Gomez-Escobar LG, Ma L, Gilani A, Alvarez-Mulett S, Sholle ET, Chandar V, Bram Y (2021) Hyperglycemia in acute COVID-19 is characterized by insulin resistance and adipose tissue infectivity by SARS-CoV-2. Cell Metab 33:2174–2188. https://doi.org/10.1016/j.cmet.2021.09.009
Anand VK, Alemar G, Griswold JA Jr (1992) Intracranial complications of mucormycosis: an experimental model and clinical review. Laryngoscope 102:656–662. https://doi.org/10.1288/00005537-199206000-00011
Giri B, Dey S, Das T, Sarkar M, Banerjee J, Dash SK (2018) Chronic hyperglycemia mediated physiological alteration and metabolic distortion leads to organ dysfunction, infection, cancer progression and other pathophysiological consequences: an update on glucose toxicity. Biomed Pharmacother 107:306–328. https://doi.org/10.1016/j.biopha.2018.07.157
Andrianaki AM, Kyrmizi I, Thanopoulou K et al (2018) Iron restriction inside macrophages regulates pulmonary host defense against Rhizopus species. Nat Commun 9:3333. https://doi.org/10.1038/s41467-018-05820-2
Tabassum T, Araf Y, Moin AT et al (2022) COVID-19-associated-mucormycosis: possible role of free iron uptake and immunosuppression. MolBiol Rep 49:747–754. https://doi.org/10.1007/s11033-021-06862-4
Cooter RD, Lim IS, Ellis DH, Leitch IO (1990) Burn wound zygomycosis caused by Apophysomyceselegans. J Clin Microbiol 28(9):2151–2153. https://doi.org/10.1128/jcm.28.9.2151-2153.1990
Hasanin MS, Hashem AH, Abd El-Sayed ES et al (2020) Green ecofriendly bio-deinking of mixed office waste paper using various enzymes from Rhizopusmicrosporus AH3: efficiency and characteristics. Cellulose 27:4443–4453. https://doi.org/10.1007/s10570-020-03071-3
Peraza-Reyes L, Malagnac F (2016) 16 Sexual development in fungi. In: Wendland J (eds) Growth, differentiation and sexuality. The Mycota, vol 1. Springer, Cham. https://doi.org/10.1007/978-3-319-25844-7_16
Karthaus M (2011) Prophylaxis and treatment of invasive aspergillosis with voriconazole, posaconazole and caspofungin—review of the literature. Eur J Med Res 16:145. https://doi.org/10.1186/2047-783X-16-4-145
Feys S, Gonçalves SM, Khan M, Choi S, Boeckx B, Chatelain D, Cunha C, Debaveye Y, Hermans G, Hertoghs M, Humblet-Baron S (2022) Lung epithelial and myeloid innate immunity in influenza-associated or COVID-19-associated pulmonary aspergillosis: an observational study. Lancet Respir Med 10:1147–1159. https://doi.org/10.1016/S2213-2600(22)00259-4
Chai LY, Vonk AG, Kullberg BJ, Verweij PE, Verschueren I, Van Der Meer JW, Joosten LA, Latgé JP, Netea MG (2011) Aspergillus fumigatus cell wall components differentially modulate host TLR2 and TLR4 responses. Microbes Infect 13:151–159. https://doi.org/10.1016/j.micinf.2010.10.005
Speth C, Rambach G, Lass-Flörl C, Howell PL, Sheppard DC (2019) Galactosaminogalactan (GAG) and its multiple roles in Aspergillus pathogenesis. Virulence 10:976–983. https://doi.org/10.1080/21505594.2019.1568174
Kim YM, Shin EC (2021) Type I and III interferon responses in SARS-CoV-2 infection. ExpMol Med 53:750–760. https://doi.org/10.1038/s12276-021-00592-0
Yao Y, Subedi K, Liu T et al (2022) Surface translocation of ACE2 and TMPRSS2 upon TLR4/7/8 activation is required for SARS-CoV-2 infection in circulating monocytes. Cell Discov 8:89. https://doi.org/10.1038/s41421-022-00453-8
Scioli MG, Storti G, D’Amico F, Rodríguez Guzmán R, Centofanti F, Doldo E, Céspedes Miranda EM, Orlandi A (1995) Oxidative stress and new pathogenetic mechanisms in endothelial dysfunction: potential diagnostic biomarkers and therapeutic targets. J Clin Med 2020:9. https://doi.org/10.3390/jcm9061995
Beserra DR, Alberca RW, Branco ACCC, Oliveira LM, de Souza Andrade MM, Gozzi-Silva SC, Teixeira FME, Yendo TM, da Silva Duarte AJ, Sato MN (2022) Upregulation of PD-1 expression and high sPD-L1 levels associated with COVID-19 severity. J Immunol Res. https://doi.org/10.1155/2022/9764002
Wurster S, Robinson P, Albert ND, Tarrand JJ, Goff M, Swamydas M, Lim JK, Lionakis MS, Kontoyiannis DP (2020) Protective activity of programmed cell death protein 1 blockade and synergy with caspofungin in a murine invasive pulmonary aspergillosis model. J Infect Dis 222:989–994. https://doi.org/10.1093/infdis/jiaa264
Gafa V, Remoli ME, Giacomini E, Severa M, Grillot R, Coccia EM (2010) Enhancement of anti-Aspergillus T helper type 1 response by interferon-β-conditioned dendritic cells. Immunology 131:282–288. https://doi.org/10.1111/j.1365-2567.2010.03302.x
Heitman J, Carter DA, Dyer PS, Soll DR (2014) Sexual reproduction of human fungal pathogens. Cold Spring Harbor Perspect Med. https://doi.org/10.1101/cshperspect.a019281
Sivasankarapillai VS, Pillai AM, Rahdar A, Sobha AP, Das SS, Mitropoulos AC, Mokarrar MH, Kyzas GZ (2020) On Facing the SARS-CoV-2 (COVID-19) with combination of nanomaterials and medicine: possible strategies and first challenges. Nanomaterials 10:852. https://doi.org/10.3390/nano10050852
Acknowledgements
One of the authors Dola Sundeep acknowledges the Ministry of Education, Government of India for providing financial support to conduct this study. Kovuri Umadevi acknowledges the Department of Pathology, Government Medical College and Hospital, Nizamabad for providing the slides to conduct the study.
Funding
Ministry of Education, Government of India, is acknowledged for providing financial assistance in to conduct this study.
Author information
Authors and Affiliations
Contributions
K.U: Conceptualization (equal); Data curation (lead); Formal analysis (lead); Investigation (equal); Methodology (equal); Writing-original draft (lead); Writing-review & editing (equal). D.S.: Conceptualization (equal); Methodology (equal); Formal analysis (lead); Investigation (equal); Writing-review & editing (lead). E.K.V: Investigation (equal) Methodology (equal). C.C.S.: Investigation (equal); Methodology (equal); Writing-review & editing (equal). A.S. Investigation (equal); Methodology (equal). R.N.C: Investigation (equal); Methodology (equal). A.R.V: Supervision (supporting); Writing-review & editing (supporting).
Corresponding authors
Ethics declarations
Conflict of interest
All the authors declare that we don’t have any financial or any other conflict of interests.
Ethical Approval
This study was conducted following the necessary ethical approvals, as delineated by the Institutional Ethical Committee (IEC), Government Medical College and Hospital, Nizamabad, India, under the reference ECR/1896/Inst/TG/2023. The research was undertaken in strict accordance with the ethical principles laid down in the 1964 Declaration of Helsinki and its subsequent amendments. This included an extensive review and approval by the named institutional ethics committee, ensuring adherence to established ethical norms. Any potential doubts regarding ethical standards were addressed and explicitly approved by the committee. The authors declare that the manuscript is prepared by obeying the Ethical Standards as described in the Committee on Publication Ethics (COPE).
Consent of Participation
The consent of participation was obtained a written informed consent from the patients for participation.
Consent for Publication
The consent of publication was obtained a written informed consent from the patients for participation.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Umadevi, K., Sundeep, D., Varadharaj, E.K. et al. Precision Detection of Fungal Co-Infections for Enhanced COVID-19 Treatment Strategies Using FESEM Imaging. Indian J Microbiol (2024). https://doi.org/10.1007/s12088-024-01246-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12088-024-01246-1