Abstract
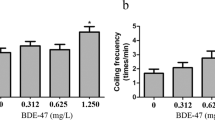
Esketamine (ESK) is the S-enantiomer of ketamine racemate (a new psychoactive substance) that can result in illusions, and alter hearing, vision, and proprioception in human and mouse. Up to now, the neurotoxicity caused by ESK at environmental level in fish is still unclear. This work studied the effects of ESK on behaviors and transcriptions of genes in dopamine and GABA pathways in zebrafish larvae at ranging from 12.4 ng L− 1 to 11141.1 ng L− 1 for 7 days post fertilization (dpf). The results showed that ESK at 12.4 ng L− 1 significantly reduced the touch response of the larvae at 48 hpf. ESK at 12.4 ng L− 1 also reduced the time and distance of larvae swimming at the outer zone during light period, which implied that ESK might potentially decrease the anxiety level of larvae. In addition, ESK increased the transcription of th, ddc, drd1a, drd3 and drd4a in dopamine pathway. Similarly, ESK raised the transcription of slc6a1b, slc6a13 and slc12a2 in GABA pathway. This study suggested that ESK could affect the heart rate and behaviors accompanying with transcriptional alterations of genes in DA and GABA pathways at early-staged zebrafish, which resulted in neurotoxicity in zebrafish larvae.



Similar content being viewed by others
References
Adams JD, Castagnoli N, Trevor AJ (1978) Quantitative analysis of ketamine enantiomers. Proc West Pharmacol Soc 21:471–472
Anacker AMJ, Moran JT, Santarelli S (2019) Enhanced social dominance and altered neuronal excitability in the prefrontal cortex of male KCC2b mutant mice. Autism Res 12(5):732–743
Baker DR, Kasprzyk-Hordern B (2011) Multi-residue analysis of drugs of abuse in wastewater and surface water by solid-phase extraction and liquid chromatography-positive electrospray ionisation tandem mass spectrometry. J Chromatogr A 1218(12):1620–1631
Baker DR, Kasprzyk-Hordern B (2013) Spatial and temporal occurrence of pharmaceuticals and illicit drugs in the aqueous environment and during wastewater treatment: new developments. Sci Total Environ 454–455:442–456
Bobo WV, Miller SC (2002) Ketamine as a preferred substance of abuse. Am J Addict 11(4):332–334
Borden LA (1996) GABA transporter heterogeneity: pharmacology and cellular localization. Neurochem Int 29(4):335–356
Borla MA, Palecek B, Budick S et al (2002) Prey capture by larval zebrafish: evidence for fine axial motor control. Brain Behav Evol 60(4):207–229
Chaudhury D, Walsh JJ, Friedman AK et al (2013) Rapid regulation of depression-related behaviors by control of midbrain dopamine neurons. Nature 493:532–536
Connaughton VP, Wetzell B, Arneson LS (2015) Elevated dopamine concentration in light-adapted zebrafish retinas is correlated with increased dopamine synthesis and metabolism. J Neurochem 135(1):101–108
Dhein S, van Koppen CJ, Brodde OE (2001) Muscarinic receptors in the mammalian heart. Pharmacol Res 44(3):161–182
Félix LM, Antunes LM, Coimbra AM (2014) Ketamine NMDA receptor-independent toxicity during zebrafish (Danio rerio) embryonic development. Neurotoxicol Teratol 41:27–34
Isomoto S, Kondo C, Takahashi N et al (1996) A novel ubiquitously distributed isoform of GIRK2 (GIRK2B) enhances GIRK1 expression of the G-protein-gated K+ current in Xenopus oocytes. Biochem Biophys Res Commun 218(1):286–291
Kenneth JL, Thomas DS (2002) Analysis of relative gene expression data using real-time quantitative PCR and the 2-∆∆CT method. Methods 25(4):402–408
Lambert AM, Bonkowsky JL, Masino MA (2012) The conserved dopaminergic diencephalospinal tract mediates vertebrate locomotor development in zebrafish larvae. J Neurosci 32(39):13488–13500
Langova V, Horka P, Hubeny J et al (2023) Ketamine disrupts locomotion and electrolocation in a novel model of schizophrenia, Gnathonemus petersii fish. J Neurosci Res 101(7):1098–1106
Lau BY, Mathur P, Gould GG et al (2011) Identification of a brain center whose activity discriminates a choice behavior in zebrafish. Proc Natl Acad Sci U S A 108(6):2581–2586
Li J, Zhang Y, Liu K et al (2018) Xiaoaiping induces developmental toxicity in zebrafish embryos through activation of ER stress, apoptosis and the wnt pathway. Front Pharmacol 9:1250
Liang XF, Zhao YQ, Liu W et al (2019) Butylated hydroxytoluene induces hyperactivity and alters dopamine-related gene expression in larval zebrafish (Danio rerio). Environ Pollut 257:113624
Mathisen LC, Skjelbred P, Skoglund LA et al (1995) Effect of ketamine, an NMDA receptor inhibitor, in acute and chronic orofacial pain. Pain 61:215–220
Motiwala Z, Aduri NG, Shaye H et al (2022) Structural basis of GABA reuptake inhibition. Nature 606:820–826
Moyon S, Frawley R, Marechal D et al (2021) TET1-mediated DNA hydroxymethylation regulates adult remyelination in mice. Nat Commun 12:3359
Oye I, Paulsen O, Maurset A (1992) Effects of ketamine on sensory perception: evidence for a role of N-methyl-D-aspartate receptors. J Pharmacol Exp Ther 260:1209–1213
Pérez-Pereira A, Ribeiro C, Teles F et al (2022) Ketamine and norketamine: enantioresolution and enantioselective aquatic ecotoxicity studies. Environ Toxicol Chem 41(3):569–579
Riehl R, Kyzar E, Allain A et al (2011) Behavioral and physiological effects of acute ketamine exposure in adult zebrafish. Neurotoxicol Teratol 33(6):658–667
Rivera C, Voipio J, Payne J et al (1999) The K+/Cl– co-transporter KCC2 renders GABA hyperpolarizing during neuronal maturation. Nature 397:251–255
Saint-Amant L, Drapeau P (1998) Time course of the development of motor behaviors in the zebrafish embryo. J Neurobiol 37(4):622–632
Schnörr SJ, Steenbergen PJ, Richardson MK (2012) Measuring thigmotaxis in larval zebrafish. Behav Brain Res 228(2):367–374
Shi WJ, Ying GG, Huang GY (2017) Transcriptional and biochemical alterations in zebrafish eleuthero-embryos (Danio rerio) after exposure to synthetic progestogen dydrogesterone. Bull Environ Contam Toxicol 99(1):39–45
Stanley KA, Curtis LR, Simonich SL et al (2009) Endosulfan I and endosulfan sulfate disrupts zebrafish embryonic development. Aquat Toxicol 95(4):355–361
Suen MFK, Chan WS, Hung KWY et al (2013) Assessments of the effects of nicotine and ketamine using tyrosine hydroxylase-green fluorescent protein transgenic zebrafish as biosensors. Biosens Bioelectron 42:177–185
Tran S, Nowicki M, Muraleetharan A et al (2015) Differential effects of dopamine D1 and D2/3 receptor antagonism on motor responses. Psychopharmacology 232(4):795–806
Tsukahara T, Masuhara M, Iwai H (2015) Repeated stress-induced expression pattern alterations of the hippocampal chloride transporters KCC2 and NKCC1 associated with behavioral abnormalities in female mice. Biochem Biophys Res Commun 465(1):145–151
Vazquez-Roig P, Andreu V, Blasco C et al (2012) Spatial distribution of illicit drugs in surface waters of the natural park of Pego-Oliva Marsh (Valencia, Spain). Environ Sci Pollut Res Int 19(4):971–982
Wang ZL, Xu ZQ, Li XQ (2019) Impacts of methamphetamine and ketamine on C. Elegans’s physiological functions at environmentally relevant concentrations and eco-risk assessment in surface waters. J Hazard Mater 363:268–276
White PF, Schüttler J, Shafer A et al (1985) Comparative pharmacology of the ketamine isomers. Studies in volunteers. Br J Anaesth 57(2):197–203
Yamada J, Okabe A, Toyoda H et al (2004) Cl– uptake promoting depolarizing GABA actions in immature rat neocortical neurones is mediated by NKCC1. J Physiol 557(3):829–841
Zakhary SM, Ayubcha D, Ansari F (2011) A behavioral and molecular analysis of ketamine in zebrafish. Synapse 65(2):160–167
Zanos P, Moaddel R, Morris PJ et al (2018) Ketamine and ketamine metabolite pharmacology: insights into therapeutic mechanisms. Pharmacol Rev 70(3):621–660
Acknowledgements
The authors would like to acknowledge the financial support from the National Natural Science Foundation of China (U22A20604, 42277268). This study is also financially funded by the Research Fund Program of Guangdong Provincial Key Laboratory of Psychoactive Substances Monitoring and Safety (2020B21201007).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no known competing financial interests or personal relationships.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, SY., Shi, WJ., Ma, DD. et al. Effects of New Psychoactive Substance Esketamine on Behaviors and Transcription of Genes in Dopamine and GABA Pathways in Zebrafish Larvae. Bull Environ Contam Toxicol 112, 51 (2024). https://doi.org/10.1007/s00128-024-03883-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00128-024-03883-0