Abstract
Invasive plants have a wide distribution worldwide. Their ability to adapt and colonize different climates is attributed to the phenotypic plasticity and the expansion of the climatic niche of the species. Simultaneously, their distribution patterns will be affected by climate change, creating or reducing the ideal areas for their establishment. We evaluated the current and potential distribution of Ulex europaeus L. in Colombia, under the global circulation models ACCESS-ESM1-5 and MIROC6 in two scenarios, for the years 2041–2060, taking into account its climate niche expansion. An ecological niche model was built for this purpose using Maxent algorithm of the KUENM, the records of the species in native and invasive condition from the GBIF portal, and records obtained through field trips and bioclimatic variables from Worldclim2 were used. Changes in potential distribution were analyzed with the Biomod2. A total of 1072 occurrence records were found for U. europaeus and 109 records were found in Colombia in the Cordillera Central and Oriental, where an expansion of the altitudinal range was evidenced from 2000 to 4052 m above sea level. Climate change models for Colombia showed that the creation of new areas for potential establishment is less than 1% and there is a reduction in potential distribution of 12 and 31% due to unsuitable climatic conditions that would affect their germination capacity. However, given its potential for adaptation, the invasions will persist in the new conditions and will continue their colonization process at higher altitudes where they find optimal temperatures for their germination and establishment.
Similar content being viewed by others
Introduction
Invasive plants are some of the most widely distributed species worldwide (Molina-Montenegro et al. 2012; Cárdenas-Cárdenas and Cortés-Peréz 2023). Their ability to disperse and colonize wide geographical areas and diverse ecosystems is attributed to the phenotypic plasticity and/or adaptive evolution of the species (Pichancourt and Van Klinken 2012; Madelón et al. 2021; Woods and Sultan 2022). In particular, the plasticity of ecologically important traits in the founding individuals of invasive species helps invasiveness by facilitating the ability to resist adverse environmental conditions or to respond positively to favorable conditions which allows the colonization of different habitats (Droste et al. 2010; Molina-Montenegro et al. 2012; Gioria et al. 2023). Once an invasive species is introduced to a place, evolution can produce genetically based phenotypic variations and adaptations which can increase resource exploitation by increasing the fitness and spread of species (Zenni et al. 2014; Dlugosch et al 2015).
Understanding niche dynamics of invasive species represents a key element of predicting future areas of invasion and their possible impacts (Barbet-Massin et al. 2018; Montagnani et al. 2022). However, the climatic niche shift in invasive plants is a widely debated topic (see Petitpierre et al. 2012; Atwater et al. 2018; Aravind et al. 2022). On the one hand, it has been shown that there are species such as Pueraria lobata (Willd.) Ohwi that show climatic niche conservatism behavior. This species, in its invasive distribution in Europe, occupies a climate range that partially overlaps with its native habitat because they had previously been adapted to a wide range of climatic conditions in its native area (Montagnani et al. 2022). Therefore, species that have broad native niches will have a better chance of establishing self-sustaining populations without the need to adapt to a new niche in the new invasive climate (Liu et al. 2020). On the other hand, some invasive species have a greater ability to expand their climatic niche because they rapidly evolve through genotype selection, genetic recombination, interspecific hybridization, and/or display specific functional traits that allow them to quickly adapt to new environments in comparison with native plants (Wan and Wang 2018; Adhikari et al. 2019; Liu et al. 2020). One example is the case documented by Broennimann et al. (2007) for Centaurea maculosa Lam., a species that has expanded its climate niche and colonized areas with climates different than its native range during its invasion in North America. Consequently, the assumption that the climatic niche of a species can be transferred between native and invasive ranges is now an issue that must be analyzed in a particular way for each species (Datta et al. 2019; Zachariah Atwater and Barney 2021).
Added to the above, the distribution patterns of invasive species are differentially affected by climate change by increasing or reducing the suitability of the habitat for their establishment (Hellmann et al. 2008; Poudel et al. 2020; Tabor and Koch 2021; Turbelin and Catford 2021). In this way, increases in CO2 concentrations and nitrogen deposition allow the establishment of viable populations and the increase in biological invasions of certain species (for example Acacia nilotica L., Indica spp., and Centaurea solstitialis L., among others) due to increased resource availability (Kriticos et al. 2003; Dukes et al. 2011; Liu et al. 2017). The opposite happens when increases in temperature and precipitation can hinder the invasive processes of plants by reducing their competitiveness due to inadequate climatic conditions (Bradley et al. 2009, 2010; Gong et al. 2020).
Currently, species distribution models (SDMs) and ecological niche models (ENM) are used to analyze species distribution patterns. In particular, the ENMs predict the geographic distributions of the species, relating records of occurrence with environmental variables which allows their use in the evaluation of niche, impacts produced by climate change and the analysis of biological invasions, among others (Cobos et al. 2019; Sillero et al. 2021). For example, these models have been used to assess current and potential global distribution changes under climate change scenarios of invasive plants such as Ligustrum lucidum W.T. Aiton and Lantana camara L., among others (Taylor et al. 2012; Montti et al. 2021).
Ulex europaeus L. is a shrub native to western Europe and the Mediterranean coasts. This species is currently considered one of the most invasive species worldwide due to its negative impacts on biodiversity, having been associated with displacing native species and the homogenization of ecosystems in areas that they have been established (León and Vargas Ríos 2009; Amaya-Villarreal and Renjifo 2010; Altamirano et al. 2016; Cárdenas-Cárdenas and Cortés-Peréz 2023). Ulex europaeus is a hexaploid species that exhibits high phenotypic plasticity in important traits for invasion such as the phenology and germination capacity of its seeds which allow it to establish itself in contrasting environmental conditions (Atlan et al. 2015; Udo et al. 2017; Muthulingam and Marambe 2022; Bellot et al. 2023). This is how U. europaeus is adapted to a wide range of climates and geographic distributions (Hill et al. 2008; Christina et al. 2020). In its native distribution area, the climate is oceanic and temperate and is found from sea level to 300 m altitude while, in an invasive condition, it is found in different latitudes (for example in Australia, North and South America and tropical islands) and at altitudes ranging from zero to 4000 m above sea level (a.s.l.) (Hornoy et al. 2011). Consequently, the ecological niche models carried out for U. europaeus by Christina et al. (2020) and Hernández‐Lambraño et al. (2017) show an expansion of the climatic niche in its invasive condition as a result of a possible adaptive evolution or product of a preadaptation to conditions that are not present in its native area. Particularly, in South America, this species suffered an expansion of its climatic niche towards places with higher temperatures, radiation and rainfall compared to its native distribution area (Hernández‐Lambraño et al. 2017).
Ulex europaeus is commonly known in Colombia as “retamo espinoso” and was introduced in the 1950s to be used as a living fence (Ocampo-Zuleta and Solorza-Bejarano 2017). However, given its invasive potential and dispersal capacity, it was distributed altitudinally from 2200 to 3700 m a.s.l., invading disturbed areas in Andean, high-Andean and paramo ecosystems and occupying extensive areas of the Cordillera Central and Oriental (Amaya-Villarreal and Renjifo 2010; Barrera-Cataño et al. 2019). In Colombia, U. europaeus affects agricultural lands, deteriorates the quality of drinking water, and generates estimated economic losses of approximately 70 million pesos per hectare associated with the cost of restoring invaded areas (Niño et al. 2018; Galappaththi et al. 2023). Given this situation, it is essential to apply strategies that anticipate and evaluate the areas susceptible to invasion for this species given that its control and eradication programs are financially expensive, wasteful, and on some occasions may be inefficient (Broadfield and McHenry 2019; Christina et al. 2020).
In this study, the current and potential distribution of U. europaeus in Colombian Andes was evaluated under climate change scenarios taking into account the expansion of the climatic niche suffered by the species. In particular, the following objectives were set: (1) To build a species niche model (ENM) considering the native climatic niche and the niche expansion experienced by U. europaeus in an invasive condition in the world and, (2) To evaluate the distribution changes of the invasive plant in climate change scenarios for Colombian Andes. For this purpose, it was hypothesized that due to the adaptive evolution and phenotypic plasticity of U. europaeus, the species will be able to adapt and migrate to different altitudes at present in the Colombian Andes, and additionally it will be able to persist successfully under climate change scenarios.
Methods
Study species
Ulex europaeus is considered one of the most invasive shrubs worldwide due to its life history traits (such as high production of long-lived seeds, rapid growth rates, long vegetative period) and the high dispersal capacity of its seeds, which can reach distances of ~ 14.65 m due to the explosive dehiscence of the pods (Altamirano et al. 2016; Broadfield and McHenry 2019; López Zea 2021; Roberts and Florentine 2021). This species mainly colonizes degraded and fragmented ecosystems that facilitate its invasion and expansion process, where it forms dense groups of individuals that negatively impact biodiversity, ecosystem services, ecological succession patterns in ecosystems, and increases the susceptibility to forest fires due to its pyrophytic character (Kitzberger et al. 2016; León Cordero et al. 2016; Osorio-Castiblanco et al. 2019).
Occurrence data
To obtain the climatic niches of U. europaeus in its native and invasive condition, the records hosted on the Global Biodiversity Information Facility (GBIF) portal with specimens preserved for the entire world were downloaded (GBIF.org 2022). Subsequently, to obtain the real distribution of the species in Colombia, a search of records was carried out in the scientific literature and geographic coordinates were requested from Colombian governmental environmental entities (Parques Nacionales Naturales and Corporaciones Autonomas Regionales). Additionally, these records were complemented with field trips that were carried out from February to December 2021 to georeferenced the populations present in the Cordillera Central in the departments of Caldas and Tolima, specifically in the buffer zone of Parque Nacional Natural Los Nevados (Table S1).
Subsequently, a spatial depuration of all the records obtained was carried out to eliminate duplicate coordinates and select only one coordinate per 1 km2 in order to avoid underestimation in the model. Likewise, a climatic depuration was carried out where the records of the species that were found in areas where the bioclimatic variables presented atypical values were removed (variables with values at zero or with very high values uncommon for the region’s climate). Also, taking into account the restricted distribution for the species in an invasive condition in Colombia, altitudinal depuration was carried out. Therefore, the records found below 2000 m a.s.l. were eliminated. Finally, random partitioning of the occurrence records was carried out to obtain a training set with 80% and an evaluation set with 20% of the remaining records for the species, as has been done with other invasive species (Chapman et al. 2019; Lakoba et al.2021).
Bioclimatic variables
The current bioclimatic and climate change variables were selected for the global circulation models ACCESS-ESM1-5 and MIROC6 with a temperature increase of 3.87 and 2.6 °C, respectively (Tatebe et al. 2019; Ziehn et al. 2020) under the scenarios of shared socioeconomic trajectories of moderate emissions (SSP245) and high emissions (SSP585) for the years 2041–2060. All bioclimatic variables were downloaded from the Worldclim2 portal (Fick and Hijmans 2017). In particular, a spatial resolution of 2.5 min was used for the records of the species in the world, and a spatial resolution of 30 s ~ 1 km2 for the projection in Colombia.
To choose the set of variables that were going to be used in the modeling, four bioclimatic variables were excluded (bio8: Mean Temperature of Wettest Quarter; bio9: Mean Temperature of Driest Quarter; bio18: Precipitation of Warmest Quarter; and bio19: Precipitation of Coldest Quarter) because by combining precipitation and temperature information into the same layer, spatial anomalies are generated, in the form of odd discontinuities between neighboring pixels (Escobar et al. 2014). Subsequently, the remaining variables were filtered by applying a Pearson coefficient (r >|0.8|) between pairs to avoid existing spatial autocorrelation. The variables that presented a correlation with absolute values for 0.8 were eliminated in order to obtain a single set with eight bioclimatic variables that represent the temperature and precipitation trends: Annual Mean Temperature (bio1), Mean Diurnal Range (bio2), Isothermality (bio3), Max Temperature of Warmest Month (bio5), Temperature Annual Range (bio7), Annual Precipitation (bio12), Precipitation of Driest Month (bio14) and Precipitation Seasonality (bio15). Also, a Non-Metric Multidimensional Scaling (NMDS) with the uncorrelated variables was performed using package Vegan (Oksanen et al. 2020), to support the climatic niche expansion suffered by the species in invasive condition.
Definition of the accessibility area (M)
The accessibility area for the species in its native and invasive distribution for all the records obtained in the world was constructed by using a 200 km buffer in the depurated records, since it covers all the presence data of the species. An accessibility area of 50 km was built (due to its distribution restricted to the mountain ranges) to assess the potential distribution under climate change scenarios in the invasive populations of Colombian Andes, so as not to overestimate the size of the populations established in the territory and the dispersal capacity of the species.
Calibration and evaluation of the model
To assess the potential distribution of the species, an ENM was carried out using the maximum entropy algorithm (Maxent) with the KUENM package (Cobos et al. 2019). To build the model, depurated records of species occurrence in both its native and invasive conditions were used, in order to represent the climatic niche expansion experienced by the species (Christina et al. 2020). Moreover, using data from both distributions provides greater accuracy in models of invasive species distribution (Broennimann and Guisan 2008). For the model, a single set of variables was used and it was calibrated by means of 15 regularization multiplier (0.1–1 with intervals of 0.1, 2–6 with intervals of 1) and the seven combinations of three feature classes (l = linear, q = quadratic and p = product). To choose the model with the highest performance and the greatest simplicity in terms of parameterization, the Area Under the Receiving Operator Curve (AUC), the omission rate (OR), and the Akaike information criterion corrected for small sample sizes were used (AICc). Finally, the model was projected onto the study area to assess the potential distribution of the species currently and under climate change scenarios using extrapolation with clamping.
Current and future potential distribution
Binary models were created from the resulting models. For this purpose, all models were reclassified taking into account a cut-off threshold of 0.8 for habitat suitability, in order to obtain the highest probability of species occurrence as suggested by Zuliani et al. (2015). Then, to assess changes in the potential distribution of U. europaeus in Colombia, the BIOMOD_RangeSize function from the Biomod2 package was used to assess areas of potential distribution stability loss or gain for this species in the future (Thuiller et al. 2016). All analyzes were performed in the R software version 4.1.2 (R Core Team 2021).
Results
In total, 1079 occurrence records were found for U. europaeus in its native distribution area and invasive condition in Europe, Africa, New Zealand, Australia and from North America to South America. Specifically, this species can be found in mean annual rainfall from 0 to 3000 mm and mean annual temperatures between 4 and 22 °C (Fig. 1). Its range of altitudinal distribution corresponds from sea level to 4052 m a.s.l. (FAUC Herbarium registration number 26740), being this last value of altitude the highest point in which the species is registered to date. This expansion in the distribution for the country was observed by this study for the Cordillera Central of Colombia in the department of Tolima.
In particular, 109 records were found located in the Cordillera Central and the Cordillera Oriental in Colombia. U. europaeus presents an altitudinal distribution from 2018 to 4052 m a.s.l., from high andean ecosystems to paramos (Fig. 2). Specifically, in the territory this species is found in the departments of Antioquia, Caldas, Cauca, Boyacá, Cundinamarca, Nariño, Norte de Santander and Tolima. Additionally, some records are found in surrounding areas or within protected areas such as Parque Nacional Natural Los Nevados, Chingaza, Sumapaz and Pisba.
A total of 105 models were obtained from the 15 regularization multiplier, seven feature classes, and a single set of variables. The final model selected with the best predictive capacity was the model with the regularization multiplier 6 and the feature class q. This model had an AUC of 1.143, a 5% omission rate of 0.076, and AICc of 27,597.12. The main variables with the highest percentage of contribution to the model were bio7 (Temperature Annual Range °C) and bio1 (Annual Mean Temperature °C) with percentages of 55.9 and 31.2, respectively.
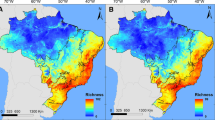
The current potential distribution model for U. europaeus covers the Cordillera Central and the Cordillera Oriental, with an altitudinal range from 2000 to altitudes above 4500 m a.s.l. (Fig. 2). Regarding the global circulation models ACCESS-ESM1-5 and MIROC6 in the SSP245 and SSP585 scenarios for U. europaeus, changes in the distribution of the species are evident with losses, gains and stable areas. In the climate change models created for Colombian Andes, the creation of new areas for potential settlement is less than 1%. On the contrary, the loss of habitat and therefore, the reduction of the potential distribution in the future fluctuates between 12 and 31% (Table 1).
Regarding the global circulation model ACCESS-ESM1-5, it presents a higher value of range of change for U. europaeus with a reduction in habitat suitability of 31 and 27% in the two scenarios due to the increase in temperature of 3.87 °C. Specifically, habitat suitability losses are evident in the Cundiboyacense highlands and in the department of Antioquia, which could lead to limiting the colonization of new areas in these zones (Fig. 3). Conversely, the reduction in habitat suitability is less in the MIROC6 global circulation model which presents an increase of 2.6 °C that will affect these areas to a lesser extent.
Discussion
In accordance with the invasive potential of U. europaeus due to the expansion of its climatic niche and its phenotypic plasticity, the results of this research provide the first projections of its current and future distribution in Colombian Andes. Specifically, ecosystems between 2000 m and 4500 + meters a.s.l. of the Cordillera Central and Oriental of Colombia are the ideal habitat for this invasive species in both present and future conditions. This is likely due to their capacity to migrate to higher altitudes, where the cold climates of the Colombian Andes favor its germination.
In particular, the ENM built for U. europaeus is explained by the bioclimatic variables related to temperature, bio1 (Annual Mean Temperature °C) and bio7 (Temperature Annual Range °C). It is clear that, although U. europaeus is adapted to a wide range of climates due to its climatic niche expansion, oceanic climates with relatively low seasonal temperature fluctuations are suitable for the species (Hornoy et al. 2011; Hernández‐Lambraño et al. 2017; Christina et al. 2020). This condition is reflected in its distribution since it responds to mean annual temperature requirements below 4 °C and above 22 °C that correspond to sites at sea level in countries with temperate regions (Atlan and Udo 2019; Christina et al. 2020). Consequently, in tropical and equatorial zones its distribution is subject to higher altitudes where it can find adequate conditions due to the presence of low temperatures (< 15 °C) which guarantee the germination of its seeds (Hill et al. 2001; Udo et al. 2017; Christina et al. 2020).
Under these criteria, the Colombian Andes are perfect habitats for the establishment of U. europaeus since they present a topographic variation with cold climates characterized by high cloudiness and low temperatures (Díaz-Granados Ortiz et al. 2005). Particularly, the ecosystems of high andean forests and paramos present in the Andes are affected by anthropogenic pressures such as processes of fragmentation, deforestation and changes in land use, aspects that make them vulnerable ecosystems for the spread of invasive species such as U. europaeus (With 2002; Calderón-Caro and Benavides 2022).
In general, this model predicts that U. europaeus will suffer loss of habitat suitability in Colombia and therefore reduction of its potential distribution, this under the global circulation models ACCESS-ESM1-5 and MIROC6 in the scenarios SSP245 and SSP585 for the years 2041–2060. Specifically, the lower loss expected in the SSP245 scenarios is due to the fact that this scenario assumes that countries will continuously reduce the intensity of energy use and move towards the sustainable development goals, the opposite of the SSP585 scenarios which assumes that the traditional economic model and the use of fossil fuels will continue to be implemented (Yang and Cui 2019).
Particularly, the decrease in habitat suitability for U. europaeus can be explained by the increase in temperature caused by climate change. This condition will reduce the germination and establishment capacity of its seeds which, consequently, will limit their dispersal process and therefore colonization or permanence in new areas (Bradley et al. 2009; Udo et al. 2017; Gong et al. 2020). It should be noted that although U. europaeus is a pyrophytic species (Kitzberger et al. 2016), the high temperatures produced in a fire act as a pre-germination treatment that helps scarification and breaks the dormancy of its seeds improving their germination rate (Hill et al. 2001; Aguilar-Garavito 2010). However, it has been shown that the optimal temperature range for U. europaeus seed germination is 0 − 15 °C, diminishing its capacity with increasing temperature (Sixtus et al. 2003; Udo et al. 2017; Broadfield and McHenry 2019). Additionally, high temperatures favor the colonization of mold that leads to the degradation of the seeds of this species (Udo et al. 2017).
This could explain the possible loss of potential distribution for the establishment of populations of U. europaeus that are evident in the departments of Antioquia, Cundinamarca, Boyacá and, in general, the reduction in their external areas present in the two models evaluated (Fig. 3). This is consistent with the fact that the alteration of climatic patterns produced by climate change will modify the distribution patterns of invasive species with losses and gains worldwide, as was demonstrated for Ligustrum lucidum (Hellmann et al. 2008; Montti et al. 2021). However, it is worth mentioning that it is possible that in other regions of Latin America the species may present gains in its potential distribution favored by climate change.
Additionally, characteristics of the species such as its hexaploidy, which allows it to express greater phenotypic plasticity and increase the possibility of adaptive evolutions (Hernández‐Lambraño et al. 2017; Christina et al. 2020; Muthulingam and Marambe 2022) can increase the adaptability of the species to a changing environment. This allows inferring that the species will continue to be present in a large proportion in the Colombian territory, affecting and/or threatening the ecosystems of paramos, high andean forests and protected areas of Colombia. This situation is considered critical because the presence of invasive species in protected areas goes against conservation objectives, causing the loss of key elements of biodiversity (Claussen and González 2015). Therefore, management and restoration plans should focus on these ecosystems that become the most vulnerable to colonization by U. europaeus due to climate change. While Resolution 0684 of 2018 in Colombia provides guidelines for the prevention and comprehensive management of U. europaeus and Genista monspessulana (L.) L.A.S Johnson (Ministerio de Ambiente y Desarrollo Sostenible 2018), it is essential to implement control activities in the buffer zones of protected areas. This is necessary to decrease the likelihood of this species invasion and mitigate its impact on the conservation objectives of these areas.
In conclusion, the analysis of current and potential distribution under climate change scenarios of invasive plants should consider the conservatism of climatic niche or the expansion that the species can experience when colonizing new exotic regions. This in order to build models more in line with the response capacity of species to changing climatic conditions in order to carry out more effective management, control or surveillance actions that help reduce the threat of global biodiversity loss produced by invasive species now and in the future.
References
Adhikari P, Jeon J-Y, Kim HW et al (2019) Potential impact of climate change on plant invasion in the Republic of Korea. J Ecology Environ 43:1–12. https://doi.org/10.1186/s41610-019-0134-3
Aguilar-Garavito M (2010) Restauración ecológica en áreas afectadas por U. europaeus L. (retamo espinoso) en la serranía el Zuque. University of Alcalá, Spain
Altamirano A, Cely JP, Etter A et al (2016) The invasive species U. europaeus (Fabaceae) shows high dynamism in a fragmented landscape of south-central Chile. Environ Monit Assess 188:1–15. https://doi.org/10.1007/s10661-016-5498-6
Amaya-Villarreal ÁM, Renjifo LM (2010) Efecto del retamo espinoso (U. europaeus) sobre las aves de borde en un bosque altoandino. Ornitología Colombiana 2010:11–25
Aravind NA, Shaanker MU, Bhat HNP et al (2022) Niche shift in invasive species: is it a case of “home away from home” or finding a “new home”? Biodivers Conserv. https://doi.org/10.1007/s10531-022-02447-0
Atlan A, Udo N (2019) The invasive niche, a multidisciplinary concept illustrated by Gorse (U. europaeus). Diversity 11:162. https://doi.org/10.3390/D11090162
Atlan A, Hornoy B, Delerue F et al (2015) Phenotypic plasticity in reproductive traits of the perennial shrub U. europaeus in response to shading: a multi-year monitoring of cultivated clones. PLoS ONE 10:e0137500. https://doi.org/10.1371/journal.pone.0137500
Atwater DZ, Ervine C, Barney JN (2018) Climatic niche shifts are common in introduced plants. Nat Ecol Evol 2:34–43. https://doi.org/10.3390/D11090162
Barbet-Massin M, Rome Q, Villemant C, Courchamp F (2018) Can species distribution models really predict the expansion of invasive species? PLoS ONE 13:e0193085. https://doi.org/10.1371/journal.pone.0193085
Barrera-Cataño JI, Contreras-Rodríguez SM, Malambo-Duarte N et al (2019) Plan de prevención, manejo y control de las especies de retamo espinoso (U. europaeus) y retamo liso (Genista monspessulana) en la jurisdicción CAR. Corporación Autónoma Regional de Cundinamarca & Pontificia Universidad Javeriana, Colombia
Bellot S, Dias PMB, Affagard M et al (2023) Molecular phylogenetics shed light on polyploid speciation in gorses (Ulex, Fabaceae: Genisteae) and on the origin of the invasive U. europaeus. Bot J Linn Soc 202:52–75. https://doi.org/10.1093/botlinnean/boac061
Bradley BA, Oppenheimer M, Wilcove DS (2009) Climate change and plant invasions: restoration opportunities ahead? Glob Chang Biol 15:1511–1521. https://doi.org/10.1111/j.1365-2486.2008.01824.x
Bradley BA, Blumenthal DM, Wilcove DS, Ziska LH (2010) Predicting plant invasions in an era of global change. Trends Ecol Evol 25:310–318. https://doi.org/10.1016/j.tree.2009.12.003
Broadfield N, McHenry MT (2019) A world of gorse: persistence of U. europaeus in managed landscapes. Plants 8:523. https://doi.org/10.3390/plants8110523
Broennimann O, Guisan A (2008) Predicting current and future biological invasions: both native and invaded ranges matter. Biol Lett 4:585–589. https://doi.org/10.1098/rsbl.2008.0254
Broennimann O, Treier UA, Müller-Schärer H et al (2007) Evidence of climatic niche shift during biological invasion. Ecol Lett 10:701–709. https://doi.org/10.1111/j.1461-0248.2007.01060.x
Calderón-Caro J, Benavides AM (2022) Deforestación y fragmentación en las áreas más biodiversas de la Cordillera Occidental de Antioquia (Colombia). Biota Colomb. https://doi.org/10.21068/2539200x.942
Cárdenas-Cárdenas MA, Cortés-Peréz F (2023) Historia de vida de U. europaeus y acciones de gestión para controlar su invasión. Actual Biol 45:1–15. https://doi.org/10.17533/udea.acbi/v45n119a03
Chapman D, Pescott OL, Roy HE, Tanner R (2019) Improving species distribution models for invasive non-native species with biologically informed pseudo-absence selection. J Biogeogr 46:1029–1040. https://doi.org/10.1111/jbi.13555
Christina M, Limbada F, Atlan A (2020) Climatic niche shift of an invasive shrub (U. europaeus): a global scale comparison in native and introduced regions. J Plant Ecol 13:42–50. https://doi.org/10.1093/JPE/RTZ041
Claussen A, González T (2015) Levantamiento de información relacionada a protocolos para el manejo de especies exóticas invasoras (EEI) en áreas protegidas continentales. Ministerio del Medio Ambiente & Programa de las Naciones Unidas para el Desarrollo (PNUD), Chile
Cobos ME, Peterson AT, Barve N, Osorio-Olvera L (2019) kuenm: an R package for detailed development of ecological niche models using Maxent. PeerJ 7:e6281. https://doi.org/10.7717/peerj.6281
Datta A, Schweiger O, Kühn I (2019) Niche expansion of the invasive plant species Ageratina adenophora despite evolutionary constraints. J Biogeogr 46:54. https://doi.org/10.1111/jbi.13579
Díaz-Granados Ortiz MA, Navarrete González JD, Suárez López T (2005) Páramos: Hidrosistemas sensibles. Revista De Ingeniería. https://doi.org/10.16924/revinge.22.8
Dlugosch KM, Alice Cang F, Barker BS et al (2015) Evolution of invasiveness through increased resource use in a vacant niche. Nat Plants 1:1–5. https://doi.org/10.1038/NPLANTS.2015.66
Droste T, Flory SL, Clay K (2010) Variation for phenotypic plasticity among populations of an invasive exotic grass. Plant Ecol 207:297–306. https://doi.org/10.1007/s11258-009-9673-5
Dukes JS, Chiariello NR, Loarie SR, Field CB (2011) Strong response of an invasive plant species (Centaurea solstitialis L.) to global environmental changes. Ecol Appl 21:1887–1894. https://doi.org/10.1890/11-0111.1
Escobar LE, Lira-Noriega A, Medina-Vogel G, Townsend Peterson A (2014) Potential for spread of the white-nose fungus (Pseudogymnoascus destructans) in the Americas: use of Maxent and Niche A to assure strict model transference. Geospat Health 9:221–229. https://doi.org/10.4081/gh.2014.19
Fick SE, Hijmans RJ (2017) WorldClim 2: new 1-km spatial resolution climate surfaces for global land areas. Int J Climatol 37:4302–4315. https://doi.org/10.1002/joc.5086
Galappaththi HSSD, de Silva WAPP, Clavijo Mccormick A (2023) A mini-review on the impact of common gorse in its introduced ranges. Trop Ecol 64:1–25. https://doi.org/10.1007/s42965-022-00239-9
GBIF.org (2022) GBIF Occurrence Download. www.gbif.org. Accessed 24 Oct 2022
Gioria M, Hulme PE, Richardson DM, Pyšek P (2023) Why are invasive plants successful? Annu Rev Plant Biol 74:635–670. https://doi.org/10.1146/annurev-arplant-070522-071021
Gong X, Chen Y, Wang T et al (2020) Double-edged effects of climate change on plant invasions: ecological niche modeling global distributions of two invasive alien plants. Sci Total Environ 740:139933. https://doi.org/10.1016/j.scitotenv.2020.139933
Hellmann JJ, Byers JE, Bierwagen BG, Dukes JS (2008) Five potential consequences of climate change for invasive species. Conserv Biol 22:534–543. https://doi.org/10.1111/j.1523-1739.2008.00951.x
Hernández-Lambraño RE, González-Moreno P, Sánchez-Agudo JÁ (2017) Towards the top: niche expansion of Taraxacum officinale and U. europaeus in mountain regions of South America. Austral Ecol 42:577–589. https://doi.org/10.1111/aec.12476
Hill RL, Gourlay AH, Barker RJ (2001) Survival of U. europaeus seeds in the soil at three sites in New Zealand. N Z J Bot 39:235–244. https://doi.org/10.1080/0028825X.2001.9512734
Hill RL, Ireson J, Sheppard AW, et al (2008) A global view of the future for biological control of gorse, U. europaeus L. In: Proceedings of the XII international symposium on biological control of weeds. CAB International, France, pp 680–686
Hornoy B, Tarayre M, Hervé M et al (2011) Invasive plants and enemy release: evolution of trait means and trait correlations in U. europaeus. PLoS ONE 6:e26275. https://doi.org/10.1371/journal.pone.0026275
Kitzberger T, Perry GLW, Paritsis J et al (2016) Fire–vegetation feedbacks and alternative states: common mechanisms of temperate forest vulnerability to fire in southern South America and New Zealand. N Z J Bot 54:247–272. https://doi.org/10.1080/0028825X.2016.1151903
Kriticos DJ, Sutherst RW, Brown JR et al (2003) Climate change and the potential distribution of an invasive alien plant: Acacia nilotica ssp. indica in Australia. J Appl Ecol 40:111–124. https://doi.org/10.1046/j.1365-2664.2003.00777.x
Lakoba VT, Atwater DZ, Thomas VE et al (2021) A global invader’s niche dynamics with intercontinental introduction, novel habitats, and climate change. Glob Ecol Conserv 31:e01848. https://doi.org/10.1016/j.gecco.2021.e01848
León O, Vargas Ríos O (2009) Caracterización espacial y conocimiento social de la invasión de retamo espinoso (U. europaeus) en la vereda El Hato (Localidad de Usme, Bogotá D.C). In: Restauración Ecológica en zonas invadidas por retamo espinoso y plantaciones forestales de especies exóticas. Universidad Nacional de Colombia, Colombia, pp 68–92
León Cordero R, Torchelsen FP, Overbeck GE, Anand M (2016) Invasive gorse (U. europaeus, Fabaceae) changes plant community structure in subtropical forest–grassland mosaics of southern Brazil. Biol Invasions 18:1629–1643. https://doi.org/10.1007/s10530-016-1106-5
Liu Y, Oduor AMO, Zhang Z et al (2017) Do invasive alien plants benefit more from global environmental change than native plants? Glob Chang Biol 23:3363–3370. https://doi.org/10.1111/gcb.13579
Liu C, Wolter C, Xian W, Jeschke JM (2020) Most invasive species largely conserve their climatic niche. Proc Natl Acad Sci U S A 117:23643–23651. https://doi.org/10.1073/pnas.2004289117
López Zea LC (2021) Caracterización de los nichos ecológicos y cambios futuros en la distribución de las especies invasoras florísticas del páramo andino. Universidad de los Andes, Spain
Madelón M, Aguirre-Acosta N, Acosta MC et al (2021) Genetic reconstruction of potential invasion pathways of Ligustrum lucidum into Argentina. Acta Oecol. https://doi.org/10.1016/j.actao.2021.103733
Ministerio de Ambiente y Desarrollo Sostenible (2018) Resolución 0684 de 2018. http://www.minambiente.gov.co/images/normativa/app/resoluciones/5b-res%20684%20de%202018.pdf. Accessed 24 Dec 2023
Molina-Montenegro MA, Penuelas J, Munné-Bosch S, Sardans J (2012) Higher plasticity in ecophysiological traits enhances the performance and invasion success of Taraxacum officinale (dandelion) in alpine environments. Biol Invasions 14:21–33. https://doi.org/10.1007/s10530-011-0055-2
Montagnani C, Casazza G, Gentili R et al (2022) Kudzu in Europe: Niche conservatism for a highly invasive plant. Biol Invasions 24:1017–1032. https://doi.org/10.1007/s10530-021-02706-1
Montti L, Velazco SJE, Travis JMJ, Grau HR (2021) Predicting current and future global distribution of invasive Ligustrum lucidum WT Aiton: assessing emerging risks to biodiversity hotspots. Divers Distrib 27:1568–1583. https://doi.org/10.1111/ddi.13303
Muthulingam P, Marambe B (2022) The invasive weed Gorse (U. europaeus L.) in Sri Lanka: implications of naturalization over a century. J Asian-Pacific Weed Sci Soc 4:21–35
Niño Y, Vega L, Ruiz B (2018) Evaluation of the energy potential of the gorse (U. europaeus) in the generation of electrical energy by gasification. Chem Eng Trans 64:481–486. https://doi.org/10.3303/CET1864081
Ocampo-Zuleta K, Solorza-Bejarano J (2017) Banco de semillas de retamo espinoso U. europaeus L. en bordes del matorral invasor en un ecosistema zonal de bosque altoandino Colombia. Biota Colomb 18:89–98. https://doi.org/10.21068/c2017.v18s01a05
Oksanen J, Blanchet FG, Friendly M, et al (2020) vegan: Community Ecology Package. 2.5–7 edn. R package
Osorio-Castiblanco DF, Peyre G, Saldarriaga JF (2019) Physicochemical analysis and essential oils extraction of the gorse (U. europaeus) and French broom (Genista monspessulana), two highly invasive species in the Colombian Andes. Sustainability 12:57. https://doi.org/10.3390/SU12010057
Petitpierre B, Kueffer C, Broennimann O et al (2012) Climatic niche shifts are rare among terrestrial plant invaders. Science 335:1344–1348. https://doi.org/10.1126/science.1215933
Pichancourt J-B, Van Klinken RD (2012) Phenotypic plasticity influences the size, shape and dynamics of the geographic distribution of an invasive plant. PLoS ONE 7:e32323. https://doi.org/10.1371/journal.pone.0032323
Poudel AS, Shrestha BB, Joshi MD et al (2020) Predicting the current and future distribution of the invasive weed Ageratina adenophora in the Chitwan-Annapurna Landscape. Nepal Mt Res Dev 40:R61. https://doi.org/10.1659/MRD-JOURNAL-D-19-00069.1
R CoreTeam, R (2021). R: A language and environment for statistical computing
Roberts J, Florentine S (2021) Biology, distribution and control of the invasive species U. europaeus (Gorse): a global synthesis of current and future management challenges and research gaps. Weed Res 61:272–281. https://doi.org/10.1111/wre.12491
Sillero N, Arenas-Castro S, Enriquez-Urzelai U et al (2021) Want to model a species niche? A step-by-step guideline on correlative ecological niche modelling. Ecol Modell 456:109671. https://doi.org/10.1016/j.ecolmodel.2021.109671
Sixtus CR, Hill GD, Scott RR (2003) The effect of temperature and scarification method on gorse (U. europaeus L) seed germination. N Z Plant Prot 56:201–205. https://doi.org/10.30843/nzpp.2003.56.6091
Tabor JA, Koch JB (2021) Ensemble models predict invasive bee habitat suitability will expand under future climate scenarios in Hawai’i. InSects 12:443. https://doi.org/10.3390/insects12050443
Tatebe H, Ogura T, Nitta T et al (2019) Description and basic evaluation of simulated mean state, internal variability, and climate sensitivity in MIROC6. Geosci Model Dev 12:2727–2765. https://doi.org/10.5194/gmd-12-2727-2019
Taylor S, Kumar L, Reid N, Kriticos DJ (2012) Climate change and the potential distribution of an invasive shrub, Lantana camara L. PLoS ONE 7:e35565. https://doi.org/10.1371/journal.pone.0035565
Thuiller W, Georges D, Engler R, Breiner F (2016) biomod2: Ensemble platform for species distribution modeling. 4.2–4 edn. R package
Turbelin A, Catford JA (2021) Invasive plants and climate change. Climate change: observed impacts on planet earth. Elsevier, UK, pp 515–532
Udo N, Tarayre M, Atlan A (2017) Evolution of germination strategy in the invasive species U. europaeus. J Plant Ecol 10:375–385. https://doi.org/10.1093/jpe/rtw032
Wan J-Z, Wang C-J (2018) Expansion risk of invasive plants in regions of high plant diversity: a global assessment using 36 species. Ecol Inform 46:8–18. https://doi.org/10.1016/j.ecoinf.2018.04.004
With KA (2002) The landscape ecology of invasive spread. Conserv Biol 16:1192–1203. https://doi.org/10.1046/j.1523-1739.2002.01064.x
Woods EC, Sultan SE (2022) Post-introduction evolution of a rapid life-history strategy in a newly invasive plant. Ecology 103:3803. https://doi.org/10.1002/ecy.3803
Yang S, Cui X (2019) Building regional sustainable development scenarios with the SSP framework. Sustainability 11:5712. https://doi.org/10.3390/su11205712
Zachariah Atwater D, Barney JN (2021) Climatic niche shifts in 815 introduced plant species affect their predicted distributions. Glob Ecol Biogeogr 30:1671–1684. https://doi.org/10.1111/geb.13342
Zenni RD, Lamy J-B, Lamarque LJ, Porté AJ (2014) Adaptive evolution and phenotypic plasticity during naturalization and spread of invasive species: implications for tree invasion biology. Biol Invasions 16:635–644. https://doi.org/10.1007/s10530-013-0607-8
Ziehn T, Chamberlain MA, Law RM et al (2020) The Australian earth system model: ACCESS-ESM1. 5. J Southern Hemisphere Earth Syst Sci 70:193–214. https://doi.org/10.1071/ES19035
Zuliani A, Massolo A, Lysyk T et al (2015) Modelling the northward expansion of Culicoides sonorensis (Diptera: Ceratopogonidae) under future climate scenarios. PLoS ONE 10:e0130294. https://doi.org/10.1371/journal.pone.0130294
Acknowledgements
We express our gratitude to the Ministerio de Ciencia, Tecnología e Innovación (MinCiencias) and Universidad de Caldas for financing the project (1127-852-71470 and 0643120): “Population status, reproductive biology and genetic diversity of the invasive plant Ulex europaeus L., in populations of the Colombian Central Cordillera” and to MCAV and EJGM as young researchers. Also, we would like to thank Parques Nacionales Naturales for the information provided and specially to park ranger Luis Carlos Villanueva for his accompaniment on field trips carried out in the department of Tolima. In addition, we express thanks to the Corporaciones Autonomas Regionales of the departments of Caldas (Corpocaldas), Cauca (CRC) and Tolima (Cortolima) for the information provided as well as to the owners of El Cedral, La Cabaña, Corrales and Alaska properties for allowing us to enter the invasions of U. europaeus present on their farms. We express our gratitude to Felipe Andrés Toro-Cardona and Andrés Miguel Betancourt for their guidance in the analysis. Finally, we thank the two anonymous reviewers for providing valuable comments and suggestions.
Funding
Open Access funding provided by Colombia Consortium. This work was financially supported by Ministerio de Ciencia, Tecnología e Innovación (MinCiencias) and Universidad de Caldas through call No. 852 of 2019 “Connecting Knowledge”.
Author information
Authors and Affiliations
Contributions
CFH, NAA, GTRR and LMAM contributed to the study conception and, CFH, NAA, GTRR and MCAV developed the study design. Data collection was carried out by MCAV with input from CFH and EJGM. Analysis was carried out by MCAV with input from CFH and NAA. The first draft of the manuscript was written by MCAV, and all authors commented on subsequent versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ángel-Vallejo, M.C., Aguirre-Acosta, N., Rodríguez-Rey, G.T. et al. Distribution models in invasive plants with climatic niche expansion: a case study of Ulex europaeus L. in Colombian Andes. Biol Invasions (2024). https://doi.org/10.1007/s10530-024-03285-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10530-024-03285-7