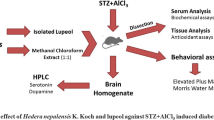
Abstract
The present study was aimed at the evaluation of neuroprotective ability of methanolic extract of Trianthema decandra (METD) against hyperglycemia-related cognitive impairment in rats. The extract of T. decandra was standardized by TLC and HPTLC methods. To verify the identity and purity of isolated compounds, they were segregated and characterized using various techniques, including UV-visible spectrophotometry, FT-IR, H-NMR, and Mass spectroscopy. α-Amylase and α-glucosidase inhibition property of the extracts were assessed in-vitro. The screening of the neuroprotective effects of METD in hyperglycemic rats was done utilizing Morri’s water (MWM) and elevated plus maze (EPM) model, as well as acetylcholinesterase (AChE) activity. The extracts of Trianthema decandra and its chemical constituents, namely quercetin and phytol, demonstrated a significant protective effect on enzymes like α-amylase and α-glucosidase. Methanol and hydroalcoholic extracts have shown the strongest inhibitory activity followed by chloroform extract. Quercetin and phytol were associated with the methanolic and chloroform extracts which were identified using TLC and HPTLC techniques. During the thirty days of the study, the induction of diabetes in the rats exhibited persistent hyperglycemia, hyperlipidemia, higher escape latency during training trials and reduced time spent in target quadrant in probe trial in Morris water maze test, and increased escape latency in EPM task. Regimen of METD (200 and 400 mg/kg) in the diabetic rats reduced the glucose levels in blood, lipid, and liver profile and showed positive results on Morri’s water and elevated plus maze tasks. During the investigation, it was determined that Trianthema decandra extracts and the chemical constituent’s quercetin and phytol in it had anti-diabetic and neuroprotective activities.
Graphical Abstract






























Similar content being viewed by others
Data Availability
No datasets were generated or analysed during the current study.
References
Alam MM, Meerza D, Naseem I (2014) Protective effect of quercetin on hyperglycemia, oxidative stress and DNA damage in alloxan induced type 2 diabetic mice. Life Sci 109:8–14. https://doi.org/10.1016/j.lfs.2014.06.005
Almermesh MH (2015) The link between type 3 diabetes and Alzheimer's disease: mechanisms of a neuro-endocrine disorder. Dissertation, Massachusetts College of Pharmacy and Health Sciences
Ardanaz CG, Ramírez MJ, Solas M (2022) Brain metabolic alterations in Alzheimer’s disease. Int J Mol Sci 23:3785. https://doi.org/10.3390/ijms23073785
Arnold SE, Arvanitakis Z, Macauley-Rambach SL, Koenig AM, Wang HY, Ahima RS, Craft S, Gandy S, Buettner C, Stoeckel LE, Holtzman DM (2018) Brain insulin resistance in type 2 diabetes and Alzheimer disease: concepts and conundrums. Nat Rev Neurol 14:168–81. https://doi.org/10.1038/nrneurol.2017.185
Awad N, Gagnon M, Messier C (2004) The relationship between impaired glucose tolerance, type 2 diabetes, and cognitive function. J Clin Exp Neuropsychol 26:1044–80. https://doi.org/10.1080/13803390490514875
Bajaj M, DeFronzo RA (2003) Metabolic and molecular basis of insulin resistance. J Nucl Cardiol 10:311–23. https://doi.org/10.1016/S1071-3581(03)00520-8
Ballard CG, Greig NH, Guillozet-Bongaarts AL, Enz A, Darvesh S (2005) Cholinesterases: roles in the brain during health and disease. Curr Alzheimer Res 2:307–18. https://doi.org/10.2174/1567205054367838
Berlanga-Acosta J, Guillén-Nieto G, Rodríguez-Rodríguez N, Bringas-Vega ML, García-del-Barco-Herrera D, Berlanga-Saez JO, García-Ojalvo A, Valdés-Sosa MJ, Valdés-Sosa PA (2020) Insulin resistance at the crossroad of Alzheimer disease pathology: a review. Front Endocrinol 11:560375. https://doi.org/10.3389/fendo.2020.560375
Bhutada P, Mundhada Y, Bansod K, Bhutada C, Tawari S, Dixit P, Mundhada D (2010) Ameliorative effect of quercetin on memory dysfunction in streptozotocin-induced diabetic rats. Neurobiol Learn Memory 94:293–302. https://doi.org/10.1016/j.nlm.2010.06.008
Blázquez E, Velázquez E, Hurtado-Carneiro V, Ruiz-Albusac JM (2014) Insulin in the brain: its pathophysiological implications for States related with central insulin resistance, type 2 diabetes and Alzheimer’s disease. Front Endocrinol 5:161. https://doi.org/10.3389/fendo.2014.00161
Daulatzai MA (2017) Cerebral hypoperfusion and glucose hypometabolism: key pathophysiological modulators promote neurodegeneration, cognitive impairment, and Alzheimer’s disease. J Neurosci Res 95:943–72. https://doi.org/10.1002/jnr.23777
Dhingra D, Kumar V (2012) Memory-enhancing activity of palmatine in mice using elevated plus maze and morris water maze. Adv Pharmacol Sci 2012(357368):1–7
Diehl T, Mullins R, Kapogiannis D (2017) Insulin resistance in Alzheimer’s disease. Transl Res 183:26–40. https://doi.org/10.1016/j.trsl.2016.12.005
El Khoury NB, Gratuze M, Papon MA, Bretteville A, Planel E (2014) Insulin dysfunction and Tau pathology. Front Cell Neurosci 8:22. https://doi.org/10.3389/fncel.2014.00022
Elmazar MM, El-Abhar HS, Schaalan MF, Farag NA (2013) Phytol/Phytanic acid and insulin resistance: potential role of phytanic acid proven by docking simulation and modulation of biochemical alterations. PLoS ONE 8:e45638. https://doi.org/10.1371/journal.pone.0045638
Fauzi A, Thoe ES, Quan TY, Yin AC (2023) Insights from insulin resistance pathways: Therapeutic approaches against Alzheimer associated diabetes mellitus. J Diabetes Complicat 10:8629. https://doi.org/10.1016/j.jdiacomp.2023.108629
Frisardi V, Solfrizzi V, Seripa D, Capurso C, Santamato A, Sancarlo D, Vendemiale G, Pilotto A, Panza F (2010) Metabolic-cognitive syndrome: a cross-talk between metabolic syndrome and Alzheimer’s disease. Age Res Rev 9:399–417. https://doi.org/10.1016/j.arr.2010.04.007
Gabbouj S, Ryhänen S, Marttinen M, Wittrahm R, Takalo M, Kemppainen S, Martiskainen H, Tanila H, Haapasalo A, Hiltunen M, Natunen T (2019) Altered insulin signaling in Alzheimer’s disease brain—special emphasis on PI3K-Akt pathway. Front Neurosci 13:629. https://doi.org/10.3389/fnins.2019.00629
Gaspar JM, Baptista FI, Macedo MP, Ambrosio AF (2016) Inside the diabetic brain: role of different players involved in cognitive decline. ACS Chem Neurosci 7:131–42. https://doi.org/10.1021/acschemneuro.5b00240
Geijselaers SL, Aalten P, Ramakers IH, De Deyn PP, Heijboer AC, Koek HL, OldeRikkert MG, Papma JM, Reesink FE, Smits LL, Stehouwer CD (2018) Association of cerebrospinal fluid (CSF) insulin with cognitive performance and CSF biomarkers of Alzheimer’s disease. J Alzheimer’s Dis 61:309–20. https://doi.org/10.3233/JAD-170522
Gil-Bea FJ, Solas M, Solomon A, Mugueta C, Winblad B, Kivipelto M, Ramirez MJ, Cedazo-Mínguez A (2010) Insulin levels are decreased in the cerebrospinal fluid of women with prodomal Alzheimer’s disease. J Alzheimer’s Dis 22:405–13. https://doi.org/10.3233/JAD-2010-100795
Goyal SN, Reddy NM, Patil KR, Nakhate KT, Ojha S, Patil CR, Agrawal YO (2016) Challenges and issues with streptozotocin-induced diabetes—a clinically relevant animal model to understand the diabetes pathogenesis and evaluate therapeutics. Chemico-Biol Interact 244:49–63. https://doi.org/10.1016/j.cbi.2015.11.032
H Ferreira-Vieira T, M Guimaraes I, R Silva F, M Ribeiro F (2016) Alzheimer’s disease: targeting the cholinergic system. Curr Neuropharmacol 14:101–15
Halim M, Halim A (2019) The effects of inflammation, aging and oxidative stress on the pathogenesis of diabetes mellitus (type 2 diabetes). Diabetes Metab Syndr 13:1165–72. https://doi.org/10.1016/j.dsx.2019.01.040
Hampel H, Mesulam MM, Cuello AC, Farlow MR, Giacobini E, Grossberg GT, Khachaturian AS, Vergallo A, Cavedo E, Snyder PJ, Khachaturian ZS (2018) The cholinergic system in the pathophysiology and treatment of Alzheimer’s disease. Brain 141:1917–33. https://doi.org/10.1093/brain/awy132
Hunt A, Schönknecht P, Henze M, Seidl U, Haberkorn U, Schröder J (2007) Reduced cerebral glucose metabolism in patients at risk for Alzheimer’s disease. Psychiatry Res 155:147–54. https://doi.org/10.1016/j.pscychresns.2006.12.003
Jacobs HI, Van Boxtel MP, Jolles J, Verhey FR, Uylings HB (2012) Parietal cortex matters in Alzheimer’s disease: an overview of structural, functional and metabolic findings. Neurosci Biobehav Rev 36:297–309. https://doi.org/10.1016/j.neubiorev.2011.06.009
Jellinger PS (2007) Metabolic consequences of hyperglycemia and insulin resistance. Clin Cornerstone 8:S30-42. https://doi.org/10.1016/S1098-3597(07)80019-6
Jing YH, Zhang L, Gao LP, Qi CC, Lv DD, Song YF, Yin J, Wang DG (2017) Autophagy plays beneficial effect on diabetic encephalopathy in type 2 diabetes: studies in vivo and in vitro. Neuroendocrinol Lett 38:27–37
Kshirsagar RP, Kothamasu MV, Patil MA, Reddy GB, Kumar BD, Diwan PV (2015) Geranium oil ameliorates endothelial dysfunction in high fat high sucrose diet induced metabolic complications in rats. J Funct Foods 15:284–293
Lane RM, Potkin SG, Enz A (2006) Targeting acetylcholinesterase and butyrylcholinesterase in dementia. Int J Neuropsychopharmacol 9:101–24. https://doi.org/10.1017/S1461145705005833
Matsuda H, Suzuki D, Asakura M, Ooi S, Saitoh R, Otokozawa R, Shirai T (2018) Effects of dietary phytol on glucose uptake and insulin secretion in vitro and in vivo. Food Nutr-Curr Res 1:29–37
Mosconi L (2005) Brain glucose metabolism in the early and specific diagnosis of Alzheimer’s disease: FDG-PET studies in MCI and AD. Eur J Nucl Med Mol Imaging 32:486–510. https://doi.org/10.1007/s00259-005-1762-7
OECD (2001) Guideline for Testing of Chemicals. Available at https://www.oecd.org/chemicalsafety/riskassessment/1948378.pdf. Accessed 27 March 2024
Oyedemi SO, Nwaogu G, Chukwuma CI, Adeyemi OT, Matsabisa MG, Swain SS, Aiyegoro OA (2020) Quercetin modulates hyperglycemia by improving the pancreatic antioxidant status and enzymes activities linked with glucose metabolism in type 2 diabetes model of rats: In silico studies of molecular interaction of quercetin with hexokinase and catalase. J Food Biochem 44:e13127. https://doi.org/10.1111/jfbc.13127
Ozdemir Z, Ozcelik AB, Uysal M (2019) Approaches based on cholinergic hypothesis and cholinesterase inhibitors in the treatment of alzheimer’s disease. Front Clin Drug Res-Alzheimer Disorders 8:154–90. https://doi.org/10.2174/9789811401893119080007
Patel TP, Rawal K, Bagchi AK, Akolkar G, Bernardes N, Dias DD, Gupta S, Singal PK (2016) Insulin resistance: an additional risk factor in the pathogenesis of cardiovascular disease in type 2 diabetes. Heart Fail Rev 21:11–23. https://doi.org/10.1007/s10741-015-9515-6
Rad SK, Arya A, Karimian H, Madhavan P, Rizwan F, Koshy S, Prabhu G (2018) Mechanism involved in insulin resistance via accumulation of β-amyloid and neurofibrillary tangles: link between type 2 diabetes and Alzheimer’s disease. Drug Design Dev Therapy. https://doi.org/10.2147/DDDT.S173970
Rajput MS, Sarkar PD (2017) Modulation of neuro-inflammatory condition, acetylcholinesterase and antioxidant levels by genistein attenuates diabetes associated cognitive decline in mice. Chemico-biol Interact 268:93–102. https://doi.org/10.1016/j.cbi.2017.02.021
Saltiel AR, Kahn CR (2001) Insulin signalling and the regulation of glucose and lipid metabolism. Nature 414:799–806. https://doi.org/10.1038/414799a
Samaha MM, Said E, Salem HA (2020) Modulatory role of imatinib mesylate on pancreatic β-cells’ secretory functions in an STZ rat model of diabetes mellitus. Chemico-Biol Interact 328:109197. https://doi.org/10.1016/j.cbi.2020.109197
Shen Z, Li ZY, Yu MT, Tan KL, Chen S (2023) Metabolic perspective of astrocyte dysfunction in Alzheimer’s disease and type 2 diabetes brains. Biomed Pharmacother 158:114206. https://doi.org/10.1016/j.biopha.2022.114206
Singh M, Kaur M, Kukreja H, Chugh R, Silakari O, Singh D (2013) Acetylcholinesterase inhibitors as Alzheimer therapy: from nerve toxins to neuroprotection. Eur J Med Chem 70:165–88. https://doi.org/10.1016/j.ejmech.2013.09.050
Stahl E (1969) Thin layer Chromatography: a laboratory handbook, 2nd edn. Springer, New York
Tumminia A, Vinciguerra F, Parisi M, Frittitta L (2018) Type 2 diabetes mellitus and Alzheimer’s disease: role of insulin signalling and therapeutic implications. Int J Mol Sci 19:3306. https://doi.org/10.3390/ijms19113306
Vinuesa A, Pomilio C, Gregosa A, Bentivegna M, Presa J, Bellotto M, Saravia F, Beauquis J (2021) Inflammation and insulin resistance as risk factors and potential therapeutic targets for Alzheimer’s disease. Front Neurosci 15:653651. https://doi.org/10.3389/fnins.2021.653651
Wang J, Hu X, Ai W, Zhang F, Yang K, Wang L, Zhu X, Gao P, Shu G, Jiang Q, Wang S (2017) Phytol increases adipocyte number and glucose tolerance through activation of PI3K/Akt signaling pathway in mice fed high-fat and high-fructose diet. Biochem Biophys Res Commun 489:432–8. https://doi.org/10.1016/j.bbrc.2017.05.160
Author information
Authors and Affiliations
Contributions
GKS carried out the Protocol writing, design of the study, SSM participated in the Phytochemical constituent Isolation, characterization and phytochemical evaluation and drafted the manuscript and participate the technical guidance of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Momina, S.S., Gandla, K. Flavonoid-Rich Trianthema decandra Ameliorates Cognitive Dysfunction in the Hyperglycemic Rats. Biochem Genet (2024). https://doi.org/10.1007/s10528-024-10744-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10528-024-10744-2