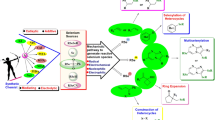
Abstract
A series of palladium (II) and platinum (II) selenolate complexes have been isolated depending upon the nature of the phosphine ligand. The substitution reaction of [PdCl2(PPh3)2] with two equivalents of NaSeC5H3(5-Me)N resulted in the hexanuclear complex [Pd6Br0.87Cl1.13{2-SeC5H3(5-Me)N2}2(PPh3)6] (1a) whereas a chelated compound [Pt{κ-Se, κ-N: SeC5H3(5-Me)N}{SeC5H3(5-Me)N}(PPh3)] (1b) has been obtained in the case of [PtCl2(PPh3)2]. However, chelated phosphine as an ancillary ligand with a similar reaction by using [PtCl2(P∩P)] (P∩P = dppm, dppe) yielded cis-configured monomer [Pt{2-SeC5H3(5-Me)N}2(P∩P)]. The trinuclear complexes [M3(µ-Se)3(dppp)3]2+ [M = Pd (2d), Pt (2e)] were isolated by reactions of [MCl2(dppp)] (M = Pd, Pt) with NaSeC5H3(5-Me)N via facile cleavage of Se-C bond in presence of chlorinated solvents. All the synthesized complexes have been characterized by elemental analyses, 1H, 31P{1H}, 77Se{1H} NMR spectroscopy. The molecular structure of complexes [Pd6Br0.87Cl1.13{2-SeC5H3(5-Me)}2(PPh3)6] (1a) and [Pt{2-SeC5H3(5-Me)N}2(dppm)].C6H5CH3 (2a) were determined by single crystal X-ray diffraction analyses.






Similar content being viewed by others
Data Availability
No datasets were generated or analysed during the current study.
References
R. Cargnelutti, R.F. Schumacher, A.L. Belladona, J.C. Kazmierczak, (2021). Coord. Chem. Rev., 426, 213537.
G. Kedarnath, V.K. Jain, (2013). Coord. Chem. Rev., 257, 1409–1435.
M. Roca Jungfer, E. Schulz Lang, U. Abram, (2022). Inorg. Chem., 61, 3785–3800.
V.K. Jain, (2020). Dalton Trans., 49, 8817–8835.
A. Arora, P. Oswal, D. Sharma, A. Tyagi, S. Purohit, P. Sharma, A. Kumar, (2022). ChemistrySelect, 7, e202201704.
P. Oswal, A. Arora, S. Gairola, A. Datta, A. Kumar, (2021). New J Chem, 4521449–21487.
S. Dey, J. Vimal, (2004). Platin. Metals Rev, 48,16–29.
J.-W. Wang, W.-J. Liu, D.-C. Zhong, T.-B. Lu, (2019). Coord. Chem. Rev., 378,237–261.
R. Oilunkaniemi, R.S. Laitinen, M. Ahlgrén, (1999). J. Organomet. Chem., 587, 200–206.
R. Oilunkaniemi, R.S. Laitinen, M. Ahlgrén, (2000). J. Organomet. Chem., 595, 232–240.
M. Risto, E.M. Jahr, M.S. Hannu-Kuure, R. Oilunkaniemi, R.S. Laitinen, (2007). J. Organomet. Chem., 692, 2193–2204.
S. Narayan, V.K. Jain, (1999). Transit. Met. Chem., 24, 409–413.
R.S. Chauhan, G. Kedarnath, A. Wadawale, A. Muñoz-Castro, R. Arratia-Perez, V.K. Jain, W. Kaim, (2010). Inorg. Chem., 49, 4179–4185.
R.S. Chauhan, G. Kedarnath, A. Wadawale, A.L. Rheingold, A. Muñoz-Castro, R. Arratia-Perez, V.K. Jain, (2012). Organometallics, 31, 1743–1750.
M. Keiji, T. Kazumitzu, I. Masato, K. Hiroshi, O. Seichi, (1998). Inorganica Chim. Acta, 281, 174–180.
C.N. Cechin, G.F. Razera, B. Tirloni, P.C. Piquini, L.M. de Carvalho, U. Abram, E.S. Lang, (2020). Inorg. Chem. Commun., 118, 107966.
S. Dey, K.V. Vivekananda, N. Bhuvanesh, (2018). Eur. J. Inorg. Chem, 2018, 3579–3586.
P. Mane, A. Pathak, N. Bhuvanesh, S. Dey, (2021). Inorg. Chem. Front., 8, 3815–3829.
G. Sheldrick, (2015). Struct. Chem., 71, 3.
C.F. Macrae, I. Sovago, S.J. Cottrell, P.T. Galek, P. McCabe, E. Pidcock, M. Platings, G.P. Shields, J.S. Stevens, M. Towler, (2020). J. Appl. Crystallogr., 53, 226–235.
M.S. Hannu-Kuure, K. Palda’n, R. Oilunkaniemi, R.S. Laitinen, M. Ahlgrén, (2003). J. Organomet. Chem., 687, 538–544.
Y. Nakatsu, Y. Nakamura, K. Matsumoto, S.i. Ooi, (1992). Inorganica Chim. Acta, 196, 81–88.
R.S. Chauhan, R.K. Sharma, G. Kedarnath, D.B. Cordes, A.M.Z. Slawin, V.K. Jain, (2012). J. Organomet. Chem., 717, 180–186.
S. Miranda, E. Cerrada, A. Luquin, A. Mendía, M. Laguna, (2012). Polyhedron, 43, 15–21.
R.S. Chauhan, G. Kedarnath, A. Wadawale, A.M.Z. Slawin, V.K. Jain, (2013). Dalton Trans., 42, 259–269.
L.B. Kumbhare, V.K. Jain, R.J. Butcher, (2006). Polyhedron, 25, 3159–3164.
V.K. Jain, L. Jain, (2005) Coord. Chem. Rev., 249, 3075–3197.
M. Capdevila, Y. Carrasco, W. Clegg, R. A. Coxall, P. González-Duarte, A. Lledós, J. Antonio Ramírez, A. Ramírez, (1999). J. Chem. Soc., Dalton Trans., 3103–3113.
R.S. Chauhan, D. Oza, S. Nigam, A. Tyagi, S. Ansari, R.J. Butcher, S. Yadav, C. Dash, (2022). J. Mol. Struct., 1248, 131368.
K. Matsumoto, C. Nishitani, M. Tadokoro, S. Okeya, (1996). J. Chem. Soc., Dalton Trans., 3335–3336.
C. Nishitani, T. Shizuka, K. Matsumoto, S. Okeya, H. Kimoto, (1998). Inorg. Chem. Commun., 1, 325–327.
A.Y. Kulkarni, S.V. Lokhande, A.A. Siddiki, R.S. Chauhan, R.J. Butcher, (2023). J. Clust. Sci.,.
R. Oilunkaniemi, R.S. Laitinen, M. Ahlgrén, (2001). J. Organomet. Chem., 623, 168–175.
R. Oilunkaniemi, R.S. Laitinen, M. Ahlgrén, (2000). J. Organomet. Chem., 595, 232–240.
J.G. Brennan, T. Siegrist, S.M. Stuczynski, M.L. Steigerwald, (1990). J. Am. Chem. Soc., 112 9233–9236.
S.S. Batsanov, (2001). Inorg. Mater., 37, 871–885.
H.P. Nayek, H. Niedermeyer, S. Dehnen, (2009). Dalton Trans., 4208–4212.
C.N. Cechin, A.V. Paz, P.C. Piquini, A.C. Bevilacqua, N.R. Pineda, N.V. Fagundes, U. Abram, E.S. Lang, B. Tirloni, (2020). Polyhedron, 177, 114315.
R.S. Chauhan, G. Kedarnath, J.A. Golen, A.L. Rheingold, V.K. Jain, (2013). J. Organomet. Chem., 723, 163–170.
S. Dey, K.V. Vivekananda, A.P. Wadawale, V.K. Jain, N. Bhuvanesh, (2017). ChemistrySelect, 2, 5073–5079.
R.S. Chauhan, G. Kedarnath, A. Wadawale, D.K. Maity, J.A. Golen, A.L. Rheingold, V.K. Jain, (2013). J. Organomet. Chem, 737, 40–46.
C.P. Morley, C.A. Webster, P. Douglas, K. Rofe, M. Di Vaira, (2010). Dalton Trans., 39, 3177–3189.
V.K. Jain, S. Kannan, R.J. Butcher, J.P. Jasinski, (1993). J. Chem. Soc., Dalton Trans., 1509–1513.
Acknowledgements
One of the authors Dr. Afsar Ali Siddiki is thankful to DST-SERB for providing grant (TARE/2022/000220) to carry out this work. Authors are also thankful to Mr. Kamlesh Sathpute from IIT Ropar for carrying out the single crystal XRD.
Funding
One of the authors Afsar Ali Siddiki is thankful to DST-SERB for providing grant (TARE/2022/000220) to carry out this work.
Author information
Authors and Affiliations
Contributions
Saili Vikram Lokhande: Investigation, methodology, data curation, writing main manuscript; Atharva Yeshwant Kulkarni: Data curation; Afsar Ali Siddiki: Data curation; Raymond J. Butcher: Data curation, writing-review and editing, Rohit Singh Chauhan: Conceptualization, supervision, writing-review and editing.
Corresponding author
Ethics declarations
Ethical Approval
Not applicable.
Competing Interests
The authors declare that they have no competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Appendix A. Supplementary Data
Appendix A. Supplementary Data
CCDC 2326944, 2294251 for complexes [Pd6Br0.87Cl1.13{2-SeC5H3(5-Me)N2}2(PPh3)6] and [Pt{2-SeC5H3(5-Me)N}2(dppm)] respectively contains the supplementary crystallographic data for this paper. These data can be obtained free of charge via http://www.ccdc.cam.ac.uk/data_request/cif, from the Cambridge Crystallographic Data Centre, 12 Union Road, Cambridge CB2 1EZ, UK; fax: (+ 44) 1223-336-033; or e-mail: deposit@ccdc.cam.ac.uk.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lokhande, S.V., Kulkarni, A.Y., Siddiki, A.A. et al. Reactivity of 5-Methylpyridyl-2-Selenolate With Platinoid Metal Precursors: Isolation of Tri and Hexanuclear Complexes via Se-Se and Se-C Bond Cleavage. J Clust Sci (2024). https://doi.org/10.1007/s10876-024-02606-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10876-024-02606-z