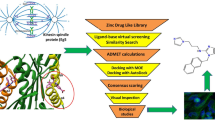
Abstract
Escherichia coli (E. coli) is a gram-negative bacterial pathogen that poses a significant clinical and epidemiologic challenge. The selection pressure brought by the insufficient use of antibiotics has resulted in the emergence of multi-drug-resistant E. coli in the past ten years. Computational and bioinformatics methods for screening inhibitors have significantly contributed to discovering novel antibacterial agents. One possible target for novel anti-virulence drugs is motility. Motility inhibitors are generally effective at concentrations lower than those required for the antibacterial properties of traditional antibiotics, and they are likely to exert less selective pressure than current medicines. Motility may be essential for bacteria to survive, find nutrients, and escape unfavorable environments and biofilm formation. The FliN is a protein forming the bulk of the C ring of the flagella and is present in multiple copies (more than 100) in bacteria. Its absence in mammals makes it an attractive drug target for drug discovery. Two-thousand seven hundred seventy-eight natural compounds from the ZINC library were screened against FliN (PDB ID: 4YXB) using PyRx AutoDock Vina, and the top compounds were selected for secondary screening after sorting the results based on their binding energy. Based on interactional analysis, binding energy (− 7.78 kcal/mol), and inhibition constant (1.98 µM), ZINC000000619481 was the best inhibitor. This compound binds exactly as per the defined active site residues of the receptor protein. Also, molecular dynamics was performed. The eigenvalue of the selected complex was 1.241657e−05. There were no ADME properties outside of the specified range for the identified hit; it fitted exactly to the binding site of the FliN receptor well and was found to be stable in MD simulation studies. Further in vitro and in vivo studies are needed to confirm its anti-bacterial activity and use as a potential antimicrobial drug against urinary tract infections caused by E. coli.








Similar content being viewed by others
References
Atanasov AG, Zotchev SB, Dirsch VM, Orhan IE, Banach M, Rollinger JM, Barreca D, Weckwerth W, Bauer R, Bayer EA, Majeed M, Bishayee A, Bochkov V, Bonn GK, Braidy N, Bucar F, Cifuentes A, D’Onofrio G, Bodkin M, Supuran CT (2021) Natural products in drug discovery: advances and opportunities. Nat Rev Drug Disc 20:200–216. https://doi.org/10.1038/s41573-020-00114-z
Brown PN, Mathews MAA, Joss LA, Hill CP, Blair DF (2005) Crystal structure of the flagellar rotor protein FliN from thermotoga maritima. J Bacteriol 187:2890–2902. https://doi.org/10.1128/JB.187.8.2890-2902.2005/SUPPL_FILE/TWO_FLIN_S.JPG
Cowan MM (1999) Plant products as antimicrobial agents. Clin Microbiol Rev 12:564. https://doi.org/10.1128/CMR.12.4.564
Daina A, Michielin O, Zoete V (2017) SwissADME: a free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci Rep 7:42717. https://doi.org/10.1038/srep42717
Dallakyan S, Olson AJ (2015) Small-molecule library screening by docking with PyRx. Mol Biol 1263:243–250. https://doi.org/10.1007/978-1-4939-2269-7
Drake D, Montie TC (1988) Flagella, motility and invasive virulence of Pseudomonas aeruginosa. J Gen Microbiol 134:43–52. https://doi.org/10.1099/00221287-134-1-43/CITE/REFWORKS
Dundas J, Ouyang Z, Tseng J, Binkowski A, Turpaz Y, Liang J (2006) CASTp: computed atlas of surface topography of proteins with structural and topographical mapping of functionally annotated residues. Nucleic Acids Research 34:116–118. https://doi.org/10.1093/nar/gkl282
Emody L, Kerényi M, Nagy G (2003) Virulence factors of uropathogenic Escherichia coli. Int J Antimicrob Agents 22:29–33. https://doi.org/10.1016/s0924-8579(03)00236-x
Flores-Mireles AL, Walker JN, Caparon M, Hultgren SJ (2015) Urinary tract infections: epidemiology, mechanisms of infection and treatment options. Nat Rev Microbiol 13:269–284. https://doi.org/10.1038/nrmicro3432
Flores-mireles AL, Walker JN, Caparon M, Hultgren SJ (2016) UTIs. Nat Rev Microbiol 13:269–284. https://doi.org/10.1038/nrmicro3432.Urinary
Gupta R, Verma R, Pradhan D, Jain AK, Umamaheswari A, Rai CS (2019) An in silico approach towards identification of novel drug targets in pathogenic species of Leptospira. PLoS ONE. https://doi.org/10.1371/JOURNAL.PONE.0221446
Islam MI, Bae JH, Ishida T, Ridone P, Lin J, Kelso MJ, Sowa Y, Buckley BJ, Baker MAB (2021) Novel amiloride derivatives that inhibit bacterial motility across multiple strains and stator types. J Bacteriol 203:367–388. https://doi.org/10.1128/JB.00367-21/SUPPL_FILE/JB.00367-21-S0003.MP4
Ivanović V, Rančić M, Arsić B, Pavlović A (2020) Lipinski’s rule of five, famous extensions and famous exceptions. Pop Sci Art 3:171–177
Josenhans C, Suerbaum S (2002) The role of motility as a virulence factor in bacteria. Int J Med Microbiol 291:605–614. https://doi.org/10.1078/1438-4221-00173
Kalia M, Singh PK, Yadav VK, Yadav BS, Sharma D, Narvi SS, Mani A, Agarwal V (2017) Structure based virtual screening for identification of potential quorum sensing inhibitors against LasR master regulator in Pseudomonas aeruginosa. Microb Pathog 107:136–143. https://doi.org/10.1016/J.MICPATH.2017.03.026
Kao CY, Lin WH, Tseng CC, Wu AB, Wang MC, Wu JJ (2014) The complex interplay among bacterial motility and virulence factors in different Escherichia coli infections. Eur J Clin Microbiol Infect Dis 33:2157–2162. https://doi.org/10.1007/S10096-014-2171-2/METRICS
Kao CY, Sheu BS, Wu JJ (2016) Helicobacter pylori infection: an overview of bacterial virulence factors and pathogenesis. Biomed J 39:14–23. https://doi.org/10.1016/J.BJ.2015.06.002
Kaper JB, Nataro JP, Mobley HL (2004) Pathogenic Escherichia coli. Nat Rev Microbiol 2:123–140. https://doi.org/10.1038/nrmicro818
Kaur H, Singh V, Kalia M, Mohan B, Taneja N (2022) Identification and functional annotation of hypothetical proteins of uropathogenic Escherichia coli strain CFT073 towards designing antimicrobial drug targets. J Biomol Struct Dyn 40:14084–14095. https://doi.org/10.1080/07391102.2021.2000499
Kaza P, Mahindroo J, Veeraraghavan B, Mavuduru RS (2019) Evaluation of risk factors for colistin resistance among uropathogenic isolates of Escherichia coli and Klebsiella Pneumoniae : a case – control study. J med microbiol. https://doi.org/10.1099/jmm.0.000986
Kelley LA, Mezulis S, Yates CM, Wass MN, Sternberg MJE (2015) The Phyre2 web portal for protein modeling, prediction and analysis. Nat Protoc 10:845–858. https://doi.org/10.1038/nprot.2015.053
Köhler CD, Dobrindt U (2011) What defines extraintestinal pathogenic Escherichia coli? Int J Med Microbiol 301:642–647. https://doi.org/10.1016/j.ijmm.2011.09.006
Kulis-Horn RK, Persicke M, Kalinowski J (2014) Histidine biosynthesis, its regulation and biotechnological application in Corynebacterium glutamicum. Microb Biotechnol 7:5–25. https://doi.org/10.1111/1751-7915.12055
Liao S, Sun A, Ojcius DM, Wu S, Zhao J, Yan J (2009) Inactivation of the FliY gene encoding a flagellar motor switch protein attenuates mobility and virulence of Leptospira interrogans strain Lai. BMC Microbiol 9:1–10. https://doi.org/10.1186/1471-2180-9-253/FIGURES/8
López-Blanco JR, Aliaga JI, Quintana-Ortí ES, Chacón P (2014) IMODS: internal coordinates normal mode analysis server. Nucleic Acids Res 42:271–276. https://doi.org/10.1093/nar/gku339
López-Blanco JR, Aliaga JI, Quintana-Ortí ES, Chacón P (2014) iMODS: internal coordinates normal mode analysis server. Nucleic Acids Res 42:271. https://doi.org/10.1093/NAR/GKU339
Maunz A, Gütlein M, Rautenberg M, Vorgrimmler D, Gebele D, Helma C, Travis KZ (2013) lazar: a modular predictive toxicology framework. Front Pharmacol 4:1–10. https://doi.org/10.3389/fphar.2013.00038
McCarter LL (2001) Polar flagellar motility of the vibrionaceae. Microbiol Mol Biol Rev 65:445–462. https://doi.org/10.1128/MMBR.65.3.445-462.2001/ASSET/12F0560B-5A64-4193-9058-A2782DEB274F/ASSETS/GRAPHIC/MR0310015007.JPEG
Navinraj S, Boopathi NM, Balasubramani V, Nakkeeran S, Raghu R, Gnanam R, Saranya N, Santhanakrishnan VP (2023) Molecular docking of nimbolide extracted from leaves of azadirachta indica with protein targets to confirm the antifungal, antibacterial and insecticidal activity. Indian J Microbiol 63:494–512. https://doi.org/10.1007/S12088-023-01104-6
O’Boyle NM, Banck M, James CA, Morley C, Vandermeersch T, Hutchison GR (2011) Open Babel: an Open chemical toolbox. J Cheminf. https://doi.org/10.1186/1758-2946-3-33
Paul K, Blair DF (2006) Organization of FliN subunits in the flagellar motor of Escherichia coli. J bacteriol 188:2502–2511. https://doi.org/10.1128/JB.188.7.2502
Peirano G, Schreckenberger PC, Pitout JDD (2011) Characteristics of NDM-1-producing Escherichia coli isolates that belong to the successful and virulent clone ST131. Antimicrob Agents Chemother 55:2986–2988. https://doi.org/10.1128/AAC.01763-10
Singhal L, Shrivastava A, Kumar N, Shekhar Sharma C (2023) Screening of Zinc database to identify MurB enzyme inhibitors for Staphylococcus aureus pathogen. Eur Chem Bull 12:11450–11463
Steffen C, Thomas K, Huniar U, Hellweg A, Rubner O, Schroer A (2010) Autodock4 and AutoDockTools4: automated docking with selective receptor flexiblity. J Comput Chem 31:2967–2970. https://doi.org/10.1002/jcc
Sterling T, Irwin JJ (2015) ZINC 15 - Ligand discovery for everyone. J Chem Inf Model 55:2324–2337. https://doi.org/10.1021/acs.jcim.5b00559
Taneja N, Sharma M (2019) Antimicrobial resistance in the environment: the Indian scenario. Indian J Med Res 149:119. https://doi.org/10.4103/ijmr.IJMR_331_18
Thomford NE, Senthebane DA, Rowe A, Munro D, Seele P, Maroyi A, Dzobo K (2018) Natural products for drug discovery in the 21st century: innovations for novel drug discovery. Int J Molecul Sci 19:1578. https://doi.org/10.3390/IJMS19061578
Wiederstein M, Sippl MJ (2007) ProSA-web: interactive web service for the recognition of errors in three-dimensional structures of proteins. Nucleic Acids Res 35:407–410. https://doi.org/10.1093/nar/gkm290
Yu Z, Li P, Merz KM (2018) Extended zinc AMBER force field (EZAFF). J Chem Theory Comput 14:242–254. https://doi.org/10.1021/acs.jctc.7b00773
Acknowledgements
We thank the Director of PGIMER Chandigarh for making this effort possible. The first author acknowledges the assistance of the ICMR, New Delhi, which is provided as a fellowship (ICMR/RA/Fellowship/2021/11125).
Funding
None.
Author information
Authors and Affiliations
Contributions
HK, and NT, Conceptualization. HK, Data curation. HK, Formal analysis. HK, Investigation. HK, Methodology. HK, Software. NT, Supervision. HK, and NT, Validation. HK, and NT Writing—original draft. HK and NT, Writing—review & editing.
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kaur, H., Taneja, N. Identification of Inhibitors for Flagellar Assembly Protein FliN of Uropathogenic Escherichia coli using Virtual Screening and Molecular Dynamics Simulation Study. Indian J Microbiol (2024). https://doi.org/10.1007/s12088-024-01252-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12088-024-01252-3