Abstract
Purpose
Boron neutron capture therapy (BNCT) is a tumor cell-selective particle-radiation therapy. In BNCT, administered p-boronophenylalanine (BPA) is selectively taken up by tumor cells, and the tumor is irradiated with thermal neutrons. High-LET α-particles and recoil 7Li, which have a path length of 5–9 μm, are generated by the capture reaction between 10B and thermal neutrons and selectively kill tumor cells that have uptaken 10B. Although BNCT has prolonged the survival time of malignant glioma patients, recurrences are still to be resolved. miRNAs, that are encapsulated in small extracellular vesicles (sEVs) in body fluids and exist stably may serve critical role in recurrence. In this study, we comprehensively investigated microRNAs (miRNAs) in sEVs released from post-BNCT glioblastoma cells.
Method
Glioblastoma U87 MG cells were treated with 25 ppm of BPA in the culture media and irradiated with thermal neutrons. After irradiation, they were plated into dishes and cultured for 3 days in the 5% CO2 incubator. Then, sEVs released into the medium were collected by column chromatography, and miRNAs in sEVs were comprehensively investigated using microarrays.
Result
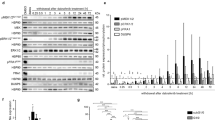
An increase in 20 individual miRNAs (ratio > 2) and a decrease in 2 individual miRNAs (ratio < 0.5) were detected in BNCT cells compared with non-irradiated cells. Among detected miRNAs, 20 miRNAs were associated with worse prognosis of glioma in Kaplan Meier Survival Analysis of overall survival in TCGA.
Conclusion
These miRNA after BNCT may proceed tumors, modulate radiation resistance, or inhibit invasion and affect the prognosis of glioma.



Similar content being viewed by others
Data availability
The datasets generated during this study are available in the Gene Expression Omnibus repository (accession number: GSE243221).
References
Weller M, van den Bent M, Preusser M et al (2021) EANO guidelines on the diagnosis and treatment of diffuse gliomas of adulthood. Nat Rev Clin Oncol 18:170–186
Barth RF, Mi P, Yang W (2018) Boron delivery agents for neutron capture therapy of cancer. Cancer Commun (Lond) 38:35
Kawabata S, Suzuki M, Hirose K et al (2021) Accelerator-based BNCT for patients with recurrent glioblastoma: a multicenter phase II study. Neurooncol Adv 3:vdab067
Kondo N, Barth RF, Miyatake SI et al (2017) Cerebrospinal fluid dissemination of high-Grade Gliomas following Boron Neutron capture Therapy occurs more frequently in the small cell subtype of IDH1(R132H) Mutation-Negative Glioblastoma. J Neurooncol 133:107–118
Steinbichler TB, Dudás J, Riechelmann H, Skvortsova II (2017) The role of sEVs in cancer metastasis. Semin Cancer Biol 44:170–181
Wubbolts R, Leckie RS, Veenhuizen PTM et al (2003) Proteomic and biochemical analyses of human B cell-derived SEVs: POTENTIAL IMPLICATIONS FOR THEIR FUNCTION AND MULTIVESICULAR BODY FORMATION*. J Biol Chem 278:10963–10972
Valadi H, Ekström K, Bossios A, Sjöstrand M, Lee JJ, Lötvall JO (2007) SEV-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol 9:654–659
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116:281–297
Valadi H, Ekström K, Bossios A, Sjöstrand M, Lee JJ, Lötvall JO (2007) Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol 9:654–659
Ohno M, Matsuzaki J, Kawauchi J et al (2019) Assessment of the diagnostic utility of serum MicroRNA classification in patients with diffuse glioma. JAMA Netw Open 2:e1916953
Kondo N, Sakurai Y, Hirota Y et al (2016) DNA damage induced by boron neutron capture therapy is partially repaired by DNA ligase IV. Radiat Environ Biophys 55:89–94
Sakurai Y, Ono K (2007) Improvement of dose distribution by central beam-shielding in boron neutron capture therapy. Phys Med Biol 52:7409–7422
Li R, Qu H, Wang S et al (2022) CancerMIRNome: an interactive analysis and visualization database for miRNome profiles of human cancer. Nucleic Acids Res 50(D1):D1139–D1146. https://doi.org/10.1093/nar/gkab784
Sun B, Pu B, Chu D, Chu X, Li W, Wei D (2013) MicroRNA-650 expression in glioma is associated with prognosis of patients. J Neurooncol 115:375–380
Chen J, Wu L, Sun Y et al (2020) Diagnostic value and clinical significance of circulating miR-650 and CA211 in detecting of gastric carcinoma. Oncol Lett 20:254
Ho KH, Chang CK, Chen PH, Wang YJ, Chang WC, Chen KC (2018) Mir-4725-3p targeting stromal interacting molecule 1 signaling is involved in xanthohumol inhibition of glioma cell invasion. J Neurochem 146:269–288
Yang XH, Guo F (2018) miR–3147 serves as an oncomiR in vulvar squamous cell cancer via Smad4 suppression. Mol Med Rep 17:6397–6404
Hao W, Zhu Y, Wang H, Guo Y (2021) miR-4270 modulates the irradiation-sensitivity of Nasopharyngeal Carcinoma Cells through modulation of p53 in vivo. Tohoku J Exp Med 254:63–70
Zou Q, Cao S (2023) miR-4270 suppresses hepatocellular carcinoma progression by inhibiting DNMT3A-mediated methylation of HGFAC promoter. PeerJ 11:e16566
Veys C, Boulouard F, Benmoussa A et al (2023) MiR-4270 acts as a tumor suppressor by directly targeting Bcl-xL in human osteosarcoma cells. Front Oncol 13:1220459
Zhang C, Liu J, Tan C et al (2016) microRNA-1827 represses MDM2 to positively regulate tumor suppressor p53 and suppress tumorigenesis. Oncotarget 7:8783–8796
Jelonek K, Widlak P, Pietrowska M (2016) The influence of Ionizing Radiation on SEV Composition, Secretion and Intercellular Communication. Protein Pept Lett 23:656–663
Abramowicz A, Wojakowska A, Marczak L et al (2019) Ionizing radiation affects the composition of the proteome of extracellular vesicles released by head-and-neck cancer cells in vitro. J Radiat Res 60:289–297
Kadhim M, Tuncay Cagatay S, Elbakrawy EM (2022) Non-targeted effects of radiation: a personal perspective on the role of sEVs in an evolving paradigm. Int J Radiat Biol 98:410–420
Arscott WT, Tandle AT, Zhao S et al (2013) Ionizing radiation and glioblastoma sEVs: implications in tumor biology and cell migration. Transl Oncol 6:638–648
Li Z, Jella KK, Jaafar L, Moreno CS, Dynan WS (2021) Characterization of sEV release and extracellular vesicle-associated miRNAs for human bronchial epithelial cells irradiated with high charge and energy ions. Life Sci Space Res 28:11–17
Acknowledgements
This work was supported by the Extramural Collaboration Research Grant of the Brain Research Institute, Niigata University to N.K., Cancer Research Institute, Kanazawa University to N.K., and Japan Society for the Promotion of Science KAKENHI Grant Number 21K09151 to N.K.
Author information
Authors and Affiliations
Contributions
Natsuko Kondo, Manabu Natsumeda and Eishu Hirata contributed to the study conception and design. Material preparation, data collection and analysis were performed by Natsuko Kondo, Tadatoshi Kinouchi, Yoshinori Sakurai and Juntaro Matsuzaki. The first draft of the manuscript was written by Natsuko Kondo and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
This work was supported by the Extramural Collaboration Research Grant of the Brain Research Institute, Niigata University to N.K., Cancer Research Institute, Kanazawa University to N.K., and Japan Society for the Promotion of Science KAKENHI Grant Number 21K09151 to N.K.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kondo, N., Kinouchi, T., Natsumeda, M. et al. Profile of miRNAs in small extracellular vesicles released from glioblastoma cells treated by boron neutron capture therapy. J Neurooncol (2024). https://doi.org/10.1007/s11060-024-04649-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11060-024-04649-8