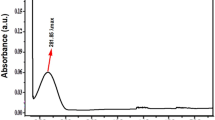
Abstract
Research for novel compounds that may block bacterial development has continued and prompted by antibiotic-resistant bacteria. The expenses of community for health care as a result of antibiotic resistance has indeed been remarkable during the last decades and demand immediate of medical attention. Consequently, this research presents the antibacterial effect of genuine metal oxide nanoparticles against Escherichia coli (E. coli) and Klebsiella pneumoniae that have been isolated from urinary tract infection patients. This is because metal oxide nanomaterials have already been utilised a compromise with some of its comprehensive implementations throughout the pharmaceutical and biological disciplines of nano-biotechnology. The biological activity of zirconium oxide (ZrO2) nanoparticles against bacteria is investigated using agar well diffusion approach. The antibacterial efficiency of nanoparticles on E. coli and Klebsiella pneumoniae using both qualitative and quantitative assessment approaches is appraised. Specifically, an aseptic technique is used to collect fifty urine samples into sterile tubes. To inoculate the patients' midstream urine on both blood agar and MacConkey agar plates, the direct streaking approach is employed. Scanning electron microscopy (SEM) and X–Ray diffraction (XRD) techniques are used to signify the physical features nanoparticle including shape and size. The identified cubic components of SEM and XRD techniques indicate the existence of ZrO2 nanoparticles and magnetic nanoparticles of particle size ranges between 5 to 50 nm. According to the data, ZrO2 nanoparticles have a bacteriostatic effect at 0.1 mg/ml with an absorption of 0.2 and a bactericidal effect at 2 mg/ml with an absorption of 0.007 on E. col isolates. Additionally, ZrO2 nanoparticles exhibit bacteriostatic (at 0.1 mg/ml with absorption of 0.3) and bactericidal (at 2 mg/ml with absorption of 0.001) effects on Klebsiella pneumoniae isolates. Among all the antibiotics utilised, gentamicin shows the lowest rate of resistance, suggesting that it is more effective against E. coli and Klebsiella pneumoniae when ZrO2 is presented.





Similar content being viewed by others
References
Mouhammed K, Gdoura R (2023) Study of the genomic characterization of antibiotic-resistant Escherichia Coli isolated from Iraqi patients with urinary tract infections. Indian J Microbiol, pp1–10.
Barišić Z, Babić-Erceg A, Borzić E, Zoranić V, Kaliterna V, Carev M (2003) Urinary tract infections in South Croatia: aetiology and antimicrobial resistance. Int J Antimicrob Agents 22:61–64
Gonzalez CM, Schaeffer AJ (1999) Treatment of urinary tract infection: what’s old, what’s new, and what works. World J Urol 17:372–382
Stamm WE, Hooton TM (1993) Management of urinary tract infections in adults. N Engl J Med 329:1328–1334
Wilson ML, Gaido L (2004) Laboratory diagnosis of urinary tract infections in adult patients. Clin Infect Dis 38:1150–1158
Sefton AM (2000) The impact of resistance on the management of urinary tract infections. Int J Antimicrob Agents 16:489–491
Bonadio M, Meini M, Spitaleri P, Gigli C (2001) Current microbiological and clinical aspects of urinary tract infections. Eur Urol 40:439–445
Wayne, P.A., 2002. National committee for clinical laboratory standards. Performance standards for antimicrobial disc susceptibility testing, 12, pp 01–53
Crude N, Tveten Y, Kristiansen BE (2001) Urinary tract infections in Norway: bacterial etiology and susceptibility. A retrospective study of clinical isolates. Clin Microbiolo Infect 7:543–547
Kripke C (2005) Duration of therapy for women with uncomplicated UTI. Am Fam Phys 72:2219
Sundqvist M, Kahlmeter G (2009) ‘Pre-emptive culturing’will improve the chance of ‘getting it right’when empirical therapy of urinary tract infections fails. J Antimicrob Chemother 64:227–228
Farajnia S, Alikhani MY, Ghotaslou R, Naghili B, Nakhlband A (2009) Causative agents and antimicrobial susceptibilities of urinary tract infections in the northwest of Iran. Int J Infect Dis 13:140–144
Atallah W, AlFuraiji N, Hussein OH (2023) Nanocomposites for prosthetic dental technology: a systemic review. J Tech 5:129–136
Sudha PN, Sangeetha K, Vijayalakshmi K, Barhoum A (2018) Nanomaterials history, classification, unique properties, production and market. In: Emerging applications of nanoparticles and architecture nanostructures. Elsevier, pp 341–384
Rajab MA, Yahiya MM, Hameed NA, Al-Obaidi MA, Al-Arkawazi RR (2024) Wear resistance of fiber-reinforced nanoparticle hybrid mixture. Int J Nanoelectron Mater (IJNeaM) 17:6–11
Gordon T, Perlstein B, Houbara O, Felner I, Banin E, Margel S (2011) Synthesis and characterization of zinc/iron oxide composite nanoparticles and their antibacterial properties. Colloids Surf, A 374:1–8
Buvaneswari K, Revathy R (2018) Biosynthesis of copper oxide nanoparticles and its antimicrobial activities. J Nanosci Technol, pp 450–451
Hu C, Sun J, Long C, Wu L, Zhou C, Zhang X (2019) Synthesis of nano zirconium oxide and its application in dentistry. Nanotechnol Rev 8:396–404
Bansal V, Rautaray D, Ahmad A, Sastry M (2004) Biosynthesis of zirconia nanoparticles using the fungus Fusarium oxysporum. J Mater Chem 14:3303–3305
Bottino A, Capannelli G, Comite A (2002) Preparation and characterization of novel porous PVDF-ZrO2 composite membranes. Desalination 146:35–40
Anu Priya G (2020) Evaluation of anti-bacterial efficacy of zirconium oxide nanoparticles (ZrO2NPs) against Streptococcus mutans and Enterococcus faecalis: an in vitro study (Doctoral dissertation, Ragas Dental College and Hospital, Chennai)
Tran TV, Nguyen DTC, Kumar PS, Din ATM, Jalil AA, Vo DVN (2022) Green synthesis of ZrO 2 nanoparticles and nanocomposites for biomedical and environmental applications: a review. Environ Chem Lett 20:1–23
Precious Ayanwale A, Reyes-López SY (2019) ZrO2–ZnO nanoparticles as antibacterial agents. ACS Omega 4:19216–19224
Gupta PK, Chauhan D, Khan ZH, Solanki PR (2020) ZrO2 nanoflowers decorated with graphene quantum dots for electrochemical immunosensing. ACS Appl Nano Mater 3:2506–2516
Sundaram R, Ramasamy G (2012) The inhibitory effect of metal oxide nanoparticles against poultry pathogens. Int J Pharm Sci Drug Res 4:157–159
Jangra SL, Stalin K, Dilbaghi N, Kumar S, Tawale J, Singh SP, Pasricha R (2012) Antimicrobial activity of zirconia (ZrO2) nanoparticles and zirconium complexes. J Nanosci Nanotechnol 12:7105–7112
Aziz SN, Al-Sallami KJ, Abd SY, Al-Musawi AMA, Mohammed MA, Abed AMH, Mohammed SQ (2019) Improving the antibacterial activity by the combination of zirconium oxide nanoparticles (ZrO2) and ceftazidime against Klebsiella pneumoniae. Global J Public Health Med 1:16–20
Ayanwale AP, Ruíz-Baltazar ADJ, Espinoza-Cristobal L, Reyes-Lopez SY (2020) Bactericidal activity study of ZrO2-Ag2O nanoparticles. Dose-Response 18:1559325820941374
Tabassum N, Kumar D, Verma D, Bohara RA, Singh MP (2021) Zirconium oxide (ZrO2) nanoparticles from antibacterial activity to cytotoxicity: a next-generation of multifunctional nanoparticles. Mater Today Commun 26:102156
Kebira AN, Ochola P, Khamadi S (2009) Isolation and antimicrobial susceptibility testing of Escherichia coli causing urinary tract infections. J Appl Biosci 22:1320–1325
Bashir M, Ashraf A, Imtiaz M, Riaz S, Naseem S (2013) Synthesis and characterization of ZrO2-ZnO nanoparticles. In: Proc. world congress on advances in nano, biomechanics, robotics and energy research (ANBRE)
Benson HJ (2002) Microbiological applications: a laboratory manual in general microbiology. McGraw-Hill, New York
Huang K, Ma H, Liu J, Huo S, Kumar A, Wei T, Zhang X, Jin S, Gan Y, Wang PC, He S (2012) Size-dependent localization and penetration of ultrasmall gold nanoparticles in cancer cells, multicellular spheroids, and tumors in vivo. ACS Nano 6:4483–4493
Hilt EE, McKinley K, Pearce MM, Rosenfeld AB, Zilliox MJ, Mueller ER, Brubaker L, Gai X, Wolfe AJ, Schreckenberger PC (2014) Urine is not sterile: use of enhanced urine culture techniques to detect resident bacterial flora in the adult female bladder. J Clin Microbiol 52:871–876
Tessema B, Kassu A, Mulu A, Yismaw G (2007) Pridominant isolates of urinary tract pathogens and their antimicrobial susceptiblity patterns in Gondar University Teaching Hospital, nothwest Ethiopia. Ethiop Med J 45:61–67
Sloan M, Farnsworth S (2006) Testing and eevaluation of nanoparticle efficacy on E. Coli, and Bacillus anthracis Spores. NSTI-Nanotech 1:595–598
Khan F, Siddiqui N, Sultan A, Rizvi M, Shukla I, Khan HM (2016) Seasonal variation in Klebsiella pneumoniae blood stream infection: a five year study. Clin Microbiol 5:247
Sepideh S, Bahri KZ, Reza PM, Reza SH, Massoud A, Roya B, Esmail NZ, Reza SA (2011) Preparation of ciprofloxacin-coated zinc oxide nanoparticles and their antibacterial effects against clinical isolates of Staphylococcus aureus and Escherichia coli. Arzneimittelforschung 61:472–476. https://doi.org/10.1055/s-0031-1296229. (PMID: 21950151)
Funding
This research received no external funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Algabar, F.A.A., Ahmed, D.S., Abbod, L.S. et al. Antibacterial Synergy: Assessing the Impact of Nano Zirconium Oxide Particles in Combination with Selected Antibiotics on Escherichia coli and Klebsiella pneumoniae Isolates from Urinary Tract Infections. Indian J Microbiol (2024). https://doi.org/10.1007/s12088-024-01271-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12088-024-01271-0