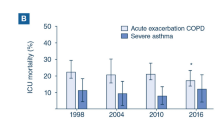
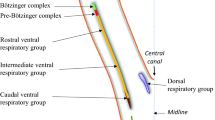
Abstract
The occurrence of major asthma symptoms is largely attributed to airway vagal hypertonia, of which the central mechanisms remain unclear. This study tests the hypotheses that endothelin-1-mediated brainstem glial activation produces asthmatic airway vagal hypertonia via enhanced action of adenosine 5’-triphosphate on neuronal purinergic P2X4 receptors. A rat model of asthma was prepared using ovalbumin. Airway vagal tone was evaluated by the recurrent laryngeal discharge and plethysmographic measurement of pulmonary function. The changes in the brainstem were examined using ELISA, Western blot, luciferin-luciferase, quantitative reverse transcription-polymerase chain reaction, enzyme activity assay and immunofluorescent staining, respectively. The results showed that in the medulla of rats, endothelin receptor type B and P2X4 receptors were primarily expressed in astrocytes and neurons, respectively, and both of which, along with endothelin-1 content, were significantly increased after ovalbumin sensitization. Ovalbumin sensitization significantly increased recurrent laryngeal discharge, which was blocked by acute intracisternal injection of P2X4 receptor antagonist 5-BDBD, knockdown of brainstem P2X4 receptors, and chronic intraperitoneal injection of endothelin receptor type B antagonist BQ788, respectively. Ovalbumin sensitization activated microglia and astrocytes and significantly decreased ecto-5’-nucleotidase activity in the medulla, and all of which, together with the increase of medullary P2X4 receptor expression and decrease of pulmonary function, were reversed by chronic BQ788 treatment. These results demonstrated that in rats, allergic airway challenge activates both microglia and astrocytes in the medulla via enhanced endothelin-1/endothelin receptor type B signaling, which subsequently causes airway vagal hypertonia via augmented adenosine 5’-triphosphate/P2X4 receptor signaling in central neurons of airway vagal reflex.
Graphical Abstract









Similar content being viewed by others
Availability of Data and Materials
All data generated or analyzed during this study are included in this article.
Abbreviations
- ATP:
-
Adenosine 5’-triphosphate
- CD73:
-
Ecto-5’-nucleotidase
- Cdyn :
-
Dynamic compliance of the lungs
- CSF:
-
Cerebral spinal fluid
- DMV:
-
Dorsal motor nucleus of the vagus
- EDNRA:
-
Endothelin receptor type A
- EDNRB:
-
Endothelin receptor type B
- ET-1:
-
Endothelin-1
- GAPDH:
-
Glyceraldehyde-3-phosphate dehydrogenase
- GFAP:
-
Glial fibrillary acidic protein
- Iba1:
-
Ionized calcium binding adapter molecule 1
- NA:
-
Nucleus ambiguus
- NTS:
-
Nucleus tractus solitarius
- P2X4R:
-
Purinergic P2X4 receptors
- OVA:
-
Ovalbumin
- Ri :
-
Inspiratory resistance of the airway
- Re :
-
Expiratory resistance of the airway
- RLD:
-
Recurrent laryngeal discharge
- RLN:
-
Recurrent laryngeal nerve
- RT-qPCR:
-
Quantitative reverse transcription-polymerase chain reaction
References
Audrit KJ, Delventhal L, Aydin Ö, Nassenstein C (2017) The nervous system of airways and its remodeling in inflammatory lung diseases. Cell Tissue Res 367:571–590
Bałuk P, Gabella G (1989) Innervation of the guinea pig trachea: a quantitative morphological study of intrinsic neurons and extrinsic nerves. J Comp Neurol 285:117–132
Caulfield JI, Schopf KJ, Cavigelli SA (2020) Peri-adolescent asthma: acute impacts on innate immune response, corticosterone, and microglia in mice. J Neuroimmunol 350:577450
Chen CY, Bonham AC, Schelegle ES, Gershwin LJ, Plopper CG, Joad JP (2001) Extended allergen exposure in asthmatic monkeys induces neuroplasticity in nucleus tractus solitarius. J Allergy Clin Immun 108:557–562
Chen X, Zeng M, He D, Yan X, Chen H, Chen Y, Xia C, Wang J, Shen L, Zhu D, Wang J (2019a) Asthmatic augmentation of airway vagal activity involves decreased central expression and activity of CD73 in rats. ACS Chem Neurosci 10:2809–2822
Chen Y, Guo Y, Yan X, Zeng M, Chen H, Qiu D, Wang J (2019b) Orexin-a excites airway vagal preganglionic neurons via activation of orexin receptor type 1 and type 2 in rats. Front Cell Neurosci 13:478
Chen Y, Li M, Liu H, Wang J (2007) The airway-related parasympathetic motoneurones in the ventrolateral medulla of newborn rats were dissociated anatomically and in functional control. Exp Physiol 92:99–108
Fukuoka T, Tokunaga A, Kondo E, Miki K, Tachibana T, Noguchi K (1998) Change in mRNAs for neuropeptides andthe GABA receptor in dorsal root ganglion neurons in a rat experimental neuropathic pain model. Pain 78:13–26
Guo Y, Yan X, Chen X, He D, Zeng M, Chen Y, Xia C, Qiu D, Wang J (2017) Corticotropin-releasing hormone modulates airway vagal preganglionic neurons of Sprague-Dawley rats at multiple synaptic sites via activation of its type 1 receptors: implications for stress-associated airway vagal excitation. Neuroscience 355:101–112
He D, Chen H, Zeng M, Xia C, Wang J, Shen L, Zhu D, Chen Y, Wang J (2020) Asthmatic Airway Vagal Hypertonia Involves Chloride Dyshomeostasis of Preganglionic Neurons in Rats. Front Neurosci-switz 14:31
Huang Y, Zhao C, Su X (2019) Neuroimmune regulation of lung infection and inflammation. Qjm-int J Med 112:483–487
Inoue K (2019) Role of the P2X4 receptor in neuropathic pain. Curr Opin Pharmacol 47:33–39
Irnaten M, Wang J, Mendelowitz D (2001) Firing properties of identified superior laryngeal neurons in the nucleus ambiguus in the rat. Neurosci Lett 303:1–4
Joos GF, Germonpré PR, Pauwels RA (2000) Neural mechanisms in asthma. Clin Exp Allergy 30(Suppl 1):60–65
Kabata H, Artis D (2019) Neuro-immune crosstalk and allergic inflammation. J Clin Invest 129:1475–1482
Knobloch J, Lin Y, Konradi J, Jungck D, Behr J, Strauch J, Stoelben E, Koch A (2013) Inflammatory responses of airway smooth muscle cells and effects of endothelin receptor antagonism. Am J Resp Cell Mol 49:114–127
Liu ZB, Song NN, Geng WY, Jin WZ, Li L, Cao YX, Qian Y, Zhu DN, Shen LL (2010) Orexin-A and respiration in a rat model of smoke-induced chronic obstructive pulmonary disease. Clin Exp Pharmacol Physiol 37:963–968
Lorenzo LE, Godin AG, Ferrini F, Bachand K, Plasencia-Fernandez I, Labrecque S, Girard AA, Boudreau D, Kianicka I, Gagnon M, Doyon N, Ribeiro-da-Silva A, De Koninck Y (2020) Enhancing neuronal chloride extrusionrescues α /α GABA -mediated analgesia in neuropathic pain. Nat Commun 11:869
Malcangio M (2019) Role of the immune system in neuropathic pain. Scand J Pain 20:33–37
Tsuda M (2016) Microglia in the spinal cord and neuropathic pain. J Diabetes Invest 7:17–26
Webster EL, Torpy DJ, Elenkov IJ, Chrousos GP (1998) Corticotropin-releasing hormone and inflammation. Ann NY Acad Sci 840:21–32
Yach D, Hawkes C, Gould CL, Hofman KJ (2004) The global burden of chronic diseases: overcoming impediments to prevention and control. J Am Med Assoc 291:2616–2622
Yamasaki R, Fujii T, Wang B, Masaki K, Kido MA, Yoshida M, Matsushita T, Kira JI (2016) Allergic inflammation leads to neuropathic pain via glial cell activation. J Neurosci 36:11929–11945
Yan X, Chen X, Guo Y, He D, Chen Y, Xia C, Wang J (2017) Arginine vasopressin alters both spontaneous and phase-locked synaptic inputs to airway vagal preganglionic neuron via activation of V(1a) receptor: insights into stress-related airway vagal excitation. Front Cell Neurosci 11:12
Zhang WJ, Zhu ZM, Liu ZX (2020) The role of P2X4 receptor in neuropathic pain and its pharmacological properties. Pharmacol Res 158:104875
Acknowledgements
This study was sponsored by the NSFC (National Natural Science Foundation of China) grants 81770003 and 81970002 to Jijiang Wang.
Funding
This study was sponsored by the NSFC (National Natural Science Foundation of China) grants 81770003 and 81970002 to Jijiang Wang.
Author information
Authors and Affiliations
Contributions
Jijiang Wang designed the study and revised the manuscript; Yun Lin wrote the main manuscript text and prepared Figs. 1–6; Tian Liu prepared Fig. 7; Hong Chen prepared Fig. 8; Ming Zeng, Shunwei Hu and Xiaoning Yu performed statistical analysis of all of the experimental data; Yonghua Chen, Chunmei Xia and Jin Wang reviewed and improved the manuscript.
Corresponding author
Ethics declarations
Ethical Approval
This study were performed in Sprague–Dawley rats. All of the animal procedures are in accordance with the recommendations of the ARRIVE guidelines, and were consented by the Ethical Committee of the Fudan University School of Basic Medical Sciences (No. 20170223–073 and No. 20190221–027).
Human and Animal Rights
Not applicable.
Competing Interests
The authors declare no competing financial interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, Y., Liu, T., Chen, H. et al. Endothelin-1-mediated Brainstem Glial Activation Produces Asthmatic Airway Vagal Hypertonia Via Enhanced ATP-P2X4 Receptor Signaling in Sprague–Dawley Rats. J Neuroimmune Pharmacol 19, 13 (2024). https://doi.org/10.1007/s11481-024-10116-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11481-024-10116-y