Abstract
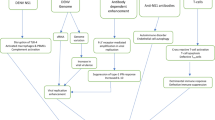
A complex interaction among virulence factors, host-genes and host immune system is considered to be responsible for dengue virus (DENV) infection and disease progression. Generation of auto-antibodies during DENV infection is a major phenomenon that plays a role in the pathophysiology of dengue hemorrhagic fever and dengue shock syndrome. Hemostasis, thrombocytopenia, hepatic endothelial dysfunction, and autoimmune blistering skin disease (pemphigus) are different clinical manifestations of dengue pathogenesis; produced due to the molecular mimicry of DENV proteins with self-antigens like coagulation factors, platelets and endothelial cell proteins. This review elaborately describes the current advancements in auto-antibody-mediated immunopathogenesis which inhibits coagulation cascade and promotes hyperfibrinolysis. Auto-antibodies like anti-endothelial cell antibodies-mediated hepatic inflammation during severe DENV infection have also been discussed. Overall, this comprehensive review provides insight to target auto-antibodies that may act as potential biomarkers for disease severity, and a ground for the development of therapeutic strategy against DENV.






Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
References
Ahmed AR, Carrozzo M, Caux F, Cirillo N, Dmochowski M, Alonso AE, Gniadecki R, Hertl M, López-Zabalza MJ, Lotti R, Pincelli C, Pittelkow M, Schmidt E, Sinha AA, Sprecher E, Grando SA (2016) Exp Dermatol 25(11):839–846. https://doi.org/10.1111/exd.13106. ‘Mono pathogenics multipathogenic explanations of pemphigus pathophysiology’
Albert DA, Talmadge DB (2010) Translational concepts in Vasculitis. Handb Cell Signal 2933–2943. https://doi.org/10.1016/B978-0-12-374145-5.00337-5
Azeredo EL, Monteiro RQ, de-Oliveira Pinto LM (2015) Thrombocytopenia in dengue: interrelationship between virus and the imbalance between coagulation and fibrinolysis and inflammatory mediators. Mediat Inflamm 1–16. https://doi.org/10.1155/2015/313842
Balakrishnan T, Bela-ong DB, Toh YX, Flamand M, Devi S, Koh MB, Hibberd ML, Ooi EE, Low JG, Leo YS et al (2011) Dengue virus activates polyreactive, natural IGG B cells after primary and secondary infection. PLoS ONE 6(12). https://doi.org/10.1371/journal.pone.0029430
Bhatt P, Sabeena SP, Varma M, Arunkumar G (2021) Current understanding of the pathogenesis of Dengue Virus infection. Curr Microbiol 78(1):17–32. https://doi.org/10.1007/s00284-020-02284-w
Broussard JA, Getsios S, Green KJ (2015) Desmosome regulation and signaling in disease. Cell Tissue Res 360(3):501–512. https://doi.org/10.1007/s00441-015-2136-5
Chad reports its first dengue outbreak (no date) CIDRAP (2023) https://www.cidrap.umn.edu/dengue/chad-reports-its-first-dengue-outbreak (Accessed: 14
Chakravarti A, Arora R, Luxemburger C (2012) Fifty years of Dengue in India. Trans R Soc Trop Med Hyg 106(5):273–282. https://doi.org/10.1016/j.trstmh.2011.12.007
Chong HY, Leow CY, Bakar AMA, Leow CH (2019) Flavivirus infection—a review of immunopathogenesis, immunological response, and immunodiagnosis. Virus Res 274:197770. https://doi.org/10.1016/j.virusres.2019.197770
Chuang YC, Lin YS, Liu CC, Liu HS, Liao SH, Shi MD, Lei HY, Yeh TM (2013) Factors contributing to the disturbance of coagulation and fibrinolysis in dengue virus infection. J Formos Med Assoc 112(1):12–17. https://doi.org/10.1016/j.jfma.2012.10.013
Chuang YC, Lin YS, Liu HS, Yeh TM (2014) Molecular mimicry between dengue virus and coagulation factors induces antibodies to inhibit thrombin activity and enhance fibrinolysis. J Virol 88(23):13759–13768. https://doi.org/10.1128/JVI.02166-14
Das S, Abreu C, Harris M, Shrader J, Sarvepalli S (2022) Severe thrombocytopenia associated with dengue fever: an evidence-based approach to management of thrombocytopenia. Case Rep Hematol 1–3. https://doi.org/10.1155/2022/3358325
Davie EW, Fujikawa K, Kisiel W (1991) The coagulation cascade: initiation, maintenance, and regulation. 30(43):10363–10370. https://doi.org/10.1021/bi00107a001
Delva E, Jennings JM, Calkins CC et al (2008) Pemphigus vulgaris IGG-induced desmoglein-3 endocytosis and desmosomal disassembly are mediated by a clathrin- and dynamin-independent mechanism. J Biol Chem 283(26):18303–18313. https://doi.org/10.1074/jbc.M710046200
Delva E, Tucker DK, Kowalczyk AP (2009) The desmosome. Cold Spring Harb Perspect Biol 1(2). https://doi.org/10.1101/cshperspect.a002543
Dengue and severe dengue. World Health Organization (2023) https://www.who.int/news-room/fact-sheets/detail/dengue-and-severe-dengue (Accessed: 14 December 2023)
Di Zenzo G, Borradori L, Muller EJ (2017) The pathogenesis of pemphigus: controversy vs complexity. Exp Dermatol 26(12):1271–1273. https://doi.org/10.1111/exd.13176
Egu DT, Schmitt T, Waschke J (2022) Mechanisms causing acantholysis in pemphigus-lessons from human skin. Front Immunol 13. https://doi.org/10.3389/fimmu.2022.884067
Farsi A, Domeneghetti MP, Brunelli T, Gori AM, Fedi S, Gensini GF, Giglioli C, Prisco D, Passaleva A, Meroni PL, Del Papa N, Abbate R (2001) Activation of the immune system and coronary artery disease: the role of anti-endothelial cell antibodies. Atherosclerosis 154(2):429–436. https://doi.org/10.1016/S0021-9150(00)00482-2
Fried JR, Gibbons RV, Kalayanarooj S, Thomas SJ, Srikiatkhachorn A et al (2010) Serotype-specific differences in the risk of Dengue hemorrhagic fever: an analysis of data collected in Bangkok, Thailand from 1994 to 2006. PLoS Negl Trop Dis 4(3). https://doi.org/10.1371/journal.pntd.0000617
Furue M, Kadono T (2017) Pemphigus, a pathomechanism of acantholysis. Australas J Dermatol 58(3):171–173. https://doi.org/10.1111/ajd.12562
Ganeshkumar P, Murhekar MV, Poornima V, Saravanakumar V, Sukumaran K et al (2018) Dengue infection in India: a systematic review and meta-analysis. PLoS Negl Trop Dis 12(7). https://doi.org/10.1371/journal.pntd.0006618
Hebbar M, Lassalle P, Delneste Y, Hatron PY, Devulder B, Tonnel AB, Janin A (2001) Activation of coagulation and fibrinolysis during dengue virus infection. J Med Virol 63(3):247–251. https://doi.org/10.1002/1096-9071(200103)63:3%3C247::aid-jmv1008%3E3.0.co;2-f
Heupel WM, Zillikens D, Drenckhahn D et al (2008) Pemphigus vulgaris IGG directly inhibit desmoglein 3-mediated transinteraction. J Immunol 181(3):1825–1834. https://doi.org/10.4049/jimmunol.181.3.1825
Hofrichter M, Dworschak J, Emtenani S, Langenhan J, Weiß F, Komorowski L et al (2018) Immunoadsorption of desmoglein-3-specific IGG abolishes the blister-inducing capacity of pemphigus vulgaris IGG in neonatal mice. Front Immunol 9. https://doi.org/10.3389/fimmu.2018.01935
Huang KJ, Li SYJ, Chen SC et al (2000) Manifestation of thrombocytopenia in dengue-2-virus-infected mice. J Gen Virol 81(9):2177–2182. https://doi.org/10.1099/0022-1317-81-9-2177
Huang Y-H et al (2001) Activation of coagulation and fibrinolysis during dengue virus infection. J Med Virol 63(3):247–251. https://doi.org/10.1002/1096-9071(200103)63:3%3C247::aid-jmv1008%3E3.0.co;2-f
Jayathilaka D, Gomes L, Jeewandara C, Jayarathna GSB, Herath D, Perera PA, Fernando S, Wijewickrama A, Hardman CS, Ogg GS et al (2018) Role of NS1 antibodies in the pathogenesis of acute secondary dengue infection. Nat Commun 9(1). https://doi.org/10.1038/s41467-018-07667-z
Jing Q, Wang M (2019) Dengue epidemiology. Global Health J 3(2):37–45. https://doi.org/10.1016/j.glohj.2019.06.002
Kakarla SG, Caminade C, Mutheneni SR, Morse AP, Upadhyayula SM et al (2019) Lag effect of climatic variables on dengue burden in India. Epidemiol Infect 147. https://doi.org/10.1017/S0950268819000608
Kamiya K, Aoyama Y, Shirafuji Y et al (2013) A higher correlation of the antibody activities against the calcium-dependent epitopes of desmoglein 3 quantified by ethylenediaminetetraacetic acid-treated enzyme-linked immunosorbent assay with clinical disease activities of pemphigus vulgaris. J Dermatol Sci 70(3):190–195. https://doi.org/10.1016/j.jdermsci.2013.02.011
Kamiya K, Aoyama Y, Wakashima C et al (2015) Atypical pemphigus with immunoglobulin G autoantibodies against Desmoglein 3 and Desmocollin 3. J Dermatol 43(4):429–431. https://doi.org/10.1111/1346-8138.13086
Koraka P, Suharti C, Setiati TE, Mairuhu ATA, Van Gorp E, Hach CE, Juffrie M, Sutaryo J, Van Der Meer GM, Groen J et al (2001) Kinetics of dengue virus-specific serum immunoglobulin classes and subclasses correlate with clinical outcome of infection. J Clin Microbiol 39(12):4332–4338. https://doi.org/10.1128/JCM.39.12.4332-4338.2001
Kouser L, Abdul-Aziz M, Nayak A, Stover CM, Sim RB, Kishore U (2013) Properdin and factor H: opposing players on the alternative complement pathway see-saw. Front Immunol 4. https://doi.org/10.3389/fimmu.2013.00093
Kurata M, Mizukawa Y, Aoyama Y, Shiohara T, Br. J (2014) Herpes simplex virus reactivation as a trigger of mucous lesions in pemphigus vulgaris. Br J Dermatol 171(3):554–560. https://doi.org/10.1111/bjd.12961
Lei HY, Yeh TM, Liu HS, Lin YS, Chen SH, Liu CC (2001) Immunopathogenesis of dengue virus infection. J Biomed Sci 8(5):377–388. https://doi.org/10.1007/BF02255946
Lin CF, Lei HY, Liu CC, Liu HS, Yeh TM, Wang ST, Yang TI, SheuFC,KuoCF,LinYS (2001) Generation of IGM anti-platelet autoantibody in dengue patients. J Med Virol 63(2):143–149. https://doi.org/10.1002/1096-9071(20000201)63:2%3C143::AID-JMV1009%3E3.0.CO;2-L
Lin SY, Lin FC, Lei YH, Liu SH, Yeh MT, Chen HS, Liu CC (2004) Antibody-mediated endothelial cell damage via nitric oxide. Curr Pharm Design 10(2):213–221. https://doi.org/10.2174/1381612043453469
Lin CF, Wan SW, Cheng HJ, Lei HY, Lin YS (2006) Autoimmune pathogenesis in dengue virus infection. Viral Immunol 19(2):127–132. https://doi.org/10.1089/vim.2006.19.127
Lin C-F, Wan S-W, Chen M-C, Lin S-C, Cheng C-C, Chiu S-C, Hsiao Y-L, Lei H-Y, Liu H-S, Yeh T-M, Yee-Shin Lin (2008) Liver injury caused by antibodies against dengue virus nonstructural protein 1 in a murine model. Lab Invest 88(10):1079–1089. https://doi.org/10.1038/labinvest.2008.70
Lin YS, Yeh TM, Lin CF, Wan SW, Chuang YC, Hsu TK et al (2011) Molecular mimicry between virus and host and its implications for dengue disease pathogenesis. Experimental Biology Med 236(5):515–523. https://doi.org/10.1258/ebm.2011.010339
Machado ARDSR, La Serra L, Turatti A, Machado AM, RoselinoAM (2017) Herpes simplex virus 1 and cytomegalovirus are associated with pemphigus vulgaris but not with pemphigus foliaceus disease. Exp Dermatol 26(10):966–968. https://doi.org/10.1111/exd.13342
Maderal AD et al (2014) Localized pemphigus foliaceus with unilateral facial involvement’, ActasDermo-Sifiliográficas. 105(4):413–417. https://doi.org/10.1016/j.ad.2013.02.014
Marchi R, Nagaswami C, Weisel JW (2009) Fibrin formation and lysis studies in dengue virus infection. Blood Coagul Fibrinolysis 20(7):575–582. https://doi.org/10.1097/MBC.0b013e32832fb1cf
Markoff LJ, Innis BL, Houghten R, Henchal LS (1991) Development of cross-reactive antibodies to plasminogen during the immune response to dengue virus infection. J Infect Dis 164(2):294–301. https://doi.org/10.1093/infdis/164.2.294
Marques-Toledo CA, Bendati MM, Codeço CT, Teixeira MM (2019) Probability of dengue transmission and propagation in a non-endemic temperate area: conceptual model and decision risk levels for early alert. Prev Control’ Parasites Vectors 12(1). https://doi.org/10.1186/s13071-018-3280-z
Martina BE, Koraka P, Osterhaus AD (2009) Dengue virus pathogenesis: an integrated view. Clin Microbiol Rev 22(4):564–581. https://doi.org/10.1128/CMR.00035-09
Miguel MC et al (2022) Autoantibodies against desmoglein 2 are not pathogenic in pemphigus. AnaisBrasileiros De Dermatologia 97(2):145–156. https://doi.org/10.1016/j.abd.2021.06.004
Mihailidou N, Katsoulas E, Panagiotou E, Farmaki A, Sklavounou H, Kiaris I, Chatzistamou (2016) Exp Dermatol 25:731
Mustafa MS, Rasotgi V, Jain S, Gupta V (2015) Discovery of fifth serotype of dengue virus (DENV-5): a New Public Health Dilemma in Dengue control. Med J Armed Forces India 71(1):67–70. https://doi.org/10.1016/j.mjafi.2014.09.011
Oishi K, Inoue S, Cinco MT, Dimaano EM, Alera MT, Alfon JA et al (2003) Correlation between increased platelet associated IgG and thrombocytopenia in secondary dengue virus infections. J Med Virol 71(2):259–264. https://doi.org/10.1002/jmv.10478
Osmani N, Labouesse M (2015) Remodeling of keratin-coupled cell adhesion complexes. Curr Opin Cell Biol 32:30–38. https://doi.org/10.1016/j.ceb.2014.10.004
Qian Y, Culton DA, Jeong JS, Trupiano N, Valenzuela JG, Diaz LA (2016) Non-infectious environmental antigens as a trigger for the initiation of an autoimmune skin disease. Autoimmun rev 15(9):923–930. https://doi.org/10.1016/j.autrev.2016.07.005
Raheel U, Faheem M, Riaz MN, Kanwal N, Javed F et al (2010) Dengue fever in the Indian subcontinent: an overview. J Infect Developing Ctries 5(04):239–247. https://doi.org/10.3855/jidc.1017
Rasizadeh R et al (2024) Viruses and thrombocytopenia. 10(6). https://doi.org/10.1016/j.heliyon.2024.e27844
Resnik N, Sepcic K, Plemenitas A et al (2011) Desmosome assembly and cell-cell adhesion are membrane raft-dependent processes. J Biol Chem 286(2):1499–1507. https://doi.org/10.1074/jbc.M110.189464
Ruocco V, Ruocco A, Lo Schiavo G, Brunetti R, Wolf (2014) Viruses and pemphigus: an intriguing never-ending story. Dermatology 229(4):310–315. https://doi.org/10.1159/000365845
Saito M, Oishi K, Inoue S, Dimaano EM, Alera MT, Alfon JA et al (2004) Association of increased platelet-associated immunoglobulins with thrombocytopenia and the severity of disease in secondary dengue virus infections. Clin Exp Immunol 138(2):299–303. https://doi.org/10.1111/j.1365-2249.2004.02626.x
Saito M, Stahley SN, Caughman (2012) Signaling dependent and independent mechanism in pemphigus vulgaris blister formation. PLoS ONE 7(12). https://doi.org/10.1371/journal.pone.0050696
Saleh MA, Hashimoto R, Kase Y et al (2015) Low pathogenicity of anti desmoglein 3 immunoglobulin G autoantibodies contributes to the atypical clinical phenotypes in pemphigus. J Dermatol 42(7):685–689. https://doi.org/10.1111/1346-8138.12888
Senger P, Sinha AA (2012) Exploring the link between herpes viruses and pemphigus vulgaris: literature review and commentary. Eur J Dermatology 22(6):728–735. https://doi.org/10.1684/ejd.2012.1836
Shih HI, Chi CY, Tsai PF, Wang YP, Chien YW (2023) Re-examination of the risk of autoimmune diseases after dengue virus infection: a population-based Cohort Study. PLoS Negl Trop Dis 17(3). https://doi.org/10.1371/journal.pntd.0011127
Song J, Park YB, Lee WK, Lee KH, Lee SK (2000) Clinical associations of anti-endothelial cell antibodies in patients with systemic lupus erythematosus. Rheumatol Int 20(1):1–7. https://doi.org/10.1007/s002960000060
Sosothikul D, Lusher JM (2005) Activation of endothelial cells, coagulation and fibrinolysis in Thai children with dengue virus infection. Blood 106(11):4039–4039. https://doi.org/10.1182/blood.V106.11.4039.4039
Spindler V, R€otzer V, Dehner C et al (2013) Peptide-mediated desmoglein 3 crosslinking prevents pemphigus vulgaris autoantibody-induced skin blistering. J Clin Invest [Preprint]. https://doi.org/10.1172/JCI60139
Srikiatkhachorn A (2009) Plasma leakage in dengue haemorrhagic fever. Thromb Haemost 102(12):1042–1049. https://doi.org/10.1160/TH09-03-0208
Srikiatkhachorn A, Mathew A, Rothman AL (2017) Immune-mediated cytokine storm and its role in severe dengue. Semin Immunopathol 39(5):563–574. https://doi.org/10.1007/s00281-017-0625-1
Stahley SN, Saito M, Faundez V et al (2014) Desmosome Assembly and disassembly are membrane raft-dependent. PLoS ONE 9(1). https://doi.org/10.1371/journal.pone.0087809
Sudipta Kumar Roy and Soumen Bhattacharjee (2021) DENV: epidemiology, biology, and disease aetiology. Can J Microbiol 67(10):687–702. https://doi.org/10.1139/cjm-2020-0572
Thomason HA, Scothern A, McHarg S et al (2010) Desmosomes: adhesive strength and signaling in health and disease. Biochem J 429(3):419–433. https://doi.org/10.1042/BJ20100567
Tracy SI, Kakalacheva K, Lunemann JD, Luzuriaga K, MiddeldorpJ,Thorley-Lawson DA (2012) Persistence of epstein-barr virus in self-reactive memory B cells. J Virol 86(22):12330–12340. https://doi.org/10.1128/jvi.01699-12
Unveiling the burden of dengue in Africa.Centers for Disease Control and Prevention https://blogs.cdc.gov/publichealthmatters/2015/07/unveiling-the-burden-of-dengue-in-africa/ (Accessed: 14 December 2023)
van der Wier G, Pas HH, Jonkman MF (2010) Experimental human cell and tissue models of pemphigus. Dermatology Res Pract 1–8. https://doi.org/10.1155/2010/143871
Vielmuth F, Waschke J, Spindler V (2015) Loss of desmoglein binding is not sufficient for keratinocyte dissociation in pemphigus. J Invest Dermatology 135(12):3068–3077. https://doi.org/10.1038/jid.2015.324
Vo HTM, Duong V, Ly S, Li QZ, Dussart P, CantaertT (2020) Autoantibody profiling in plasma of dengue virus–infected individuals. Pathogens 9(12):1060. https://doi.org/10.3390/pathogens9121060
Wan S-W, Lin C-F, Yeh T-M, Liu C-C, Liu H-S, Wang S, Ling P, Anderson R, Lei H-Y, Yee-Shin Lin (2013) Autoimmunity in dengue pathogenesis. J Formos Med Assoc 112(1):3–11. https://doi.org/10.1016/j.jfma.2012.11.006
Warter L, Appanna R, Fink K (2012) Human poly- and cross-reactive anti-viral antibodies and their impact on protection and pathology. Immunol Res 53(1–3):148–161. https://doi.org/10.1007/s12026-012-8268-8
Wunderlich J, Acuña-soto R, Alonso WJ (2017) Dengue Hospitalisations in Brazil: annual wave from west to east and recent increase among children. Epidemiol Infect 146(2):236–245. https://doi.org/10.1017/S0950268817002801
Yurasov S, Nussenzweig MC (2007) Regulation of autoreactive antibodies. Curr Opin Rheumatol 19(5):421–426. https://doi.org/10.1097/BOR.0b013e328277ef3b
Zhou Z-H, Tzioufas AG, Notkins AL (2007) Properties and function of polyreactive antibodies and polyreactive antigen-binding B cells. J Autoimmun 29(4):219–228. https://doi.org/10.1016/j.jaut.2007.07.015
Acknowledgements
The authors thankfully acknowledge Dr. Arti Soren, Officer-in-charge, Dr. Anjali Chatterjee Regional Research Institute, CCRH, Ministry of AYUSH and Dr. Subhash Kaushik, Director General, CCRH, Ministry of AYUSH, Govt. of India for their support.
Funding
This work was supported by CCRH Research grant, Ministry of AYUSH, Govt. of India (Sanction no. 17–152/2010–11/ CCRH/Tech/Virology Lab/3347).
Author information
Authors and Affiliations
Contributions
Conceptualization: Dr. Satadal Das, Dr.Avipsha Sarkar, Dr. Anirban Roy, Tanusree Ghorai Resources: Dr. Satadal Das Funding acquisition: Dr. Debadatta Nayak Visualization: Dr. Satadal Das, Tanusree Ghorai Writing - original draft: Tanusree Ghorai, Bijita Bhowmick Writing - review & editing: Dr. Satadal Das, Dr. Anirban Roy, Dr.Avipsha Sarkar, Bijita Bhowmick.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Communicated by Yusuf Akhter.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ghorai, T., Sarkar, A., Roy, A. et al. Role of auto-antibodies in the mechanisms of dengue pathogenesis and its progression: a comprehensive review. Arch Microbiol 206, 214 (2024). https://doi.org/10.1007/s00203-024-03954-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-024-03954-0