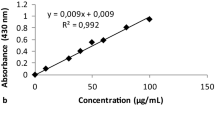
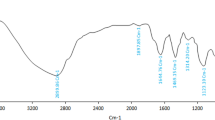
Abstract
Beta vulgaris var. crassa is undoubtedly a very important plant that is not used enough in the world. In this study, it was aimed to determine the cytotoxic activities of the components (essential oils, fatty acids, total phenol and flavonoid) found in the leaf parts of Beta vulgaris var. crassa against PC-3, MCF-7 and HeLa cancer cell lines. In addition, the effectiveness of these ingredients against bacteria and fungi that can cause serious health problems in humans was tested. In experiments, three tumor cell lines were exposed to various plant extract concentrations (31.25, 62.5, 125, 250, 500 and 1000 µg/mL) for 72 h. It was found that plant extracts showed high (SI: 2.14 > 2) cytotoxicity to PC-3 cells, moderate (SI: 1.62 < 2) to HeLa cells, and low (SI: 0.93 < 2) cytotoxicity to MCF-7 cells. Also, different plant extract concentrations were found to cause an inhibition rate of 16.3–22.3% in Staphylococcus aureus, 16.8–23.5% in Streptococcus pyogenes and 12–16.2% in Cutibacterium acnes. Similarly, inhibition rates were determined between 9.5–20.7% for Candida albicans, 3.5–7.7% for Candida auris, and 5.5–15.1% for Candida glabrata. The results showed that the plant extract exhibited a concentration-dependent cytotoxic and antimicrobial effect against both cancer cell lines and microbial pathogens.
Graphical Abstract





Similar content being viewed by others
References
Schwichtenberg K, Wenke T, Zakrzewski F, Seibt KM, Minoche A et al (2016) Diversification, evolution and methylation of short interspersed nuclear element families in sugar beet and related Amaranthaceae species. Plant J 85:229–244. https://doi.org/10.1111/tpj.13103
Hossain A, Maitra S, Pramanick B, Bhutia KL, Ahmad Z et al (2022) Wild relatives of plants as sources for the development of abiotic stress tolerance in plants. In: Plant perspectives to global climate changes. Academic Press, Cambridge, pp. 471–518. https://doi.org/10.1016/B978-0-323-85665-2.00011-X
Rozema J, Cornelisse D, Zhang Y, Li H, Bruning B et al (2015) Comparing salt tolerance of beet cultivars and their halophytic ancestor: consequences of domestication and breeding programmes. AoB Plants 7:2015. https://doi.org/10.1093/aobpla/plu083
Al-Jbawi E (2020) All about fodder beet (beta vulgaris subsp. crassa L.) as a source of forage in the world and Syria. Res J Sci 1:24–44
Thiruvengadam M, Chung IM, Samynathan R, Chandar SH, Venkidasamy B et al (2022) A comprehensive review of beetroot (Beta vulgaris L.) bioactive components in the food and pharmaceutical industries. Crit Rev Food Sci Nutr 64:708–739. https://doi.org/10.1080/10408398.2022.2108367
Slavova Y, Nenkova D, Ivanova I (2004) Optimization of nutritive medium with the purpose of rooting of fodder beet (Beta vulgaris L. var crassa) through in vitro method. Bulg J Agric Sci 10:465–468
Enchev S, Bozhanska T (2022) Chemical composition of sugar beet, fodder beet and table beet depending on the harvest period. Bulg J Agric Sci 28:1034–1039
Bhunia S, Bhowmik A, Mallick R, Mukherjee J (2021) Agronomic efficiency of animal-derived organic fertilizers and their effects on biology and fertility of soil: a review. Agronomy 11:823. https://doi.org/10.3390/agronomy11050823
Malcolm BJ, Cameron KC, Edwards GR, Di HJ, de Ruiter JM et al (2016) Nitrate leaching losses from lysimeters simulating winter grazing of fodder beet by dairy cows. New Zealand J Agric Res 59:194–203. https://doi.org/10.1080/00288233.2016.1150307
Olumese FE, Oboh HA (2016) Antioxidant and Antioxidant capacity of raw and processed Nigerian Beetroot (Beta vulgaris). Nig J Basic Appl Sci 24:35–40. https://doi.org/10.4314/njbas.v24i1.6
Kale R, Sawate AR, Kshirsagar R, Patil B, Mane R (2018) Studies on evaluation of physical and chemical composition of beetroot (Beta vulgaris L). Int J Chem Stud 6:2977–2979
Clifford T, Howatson G, West DJ, Stevenson EJ (2015) The potential benefits of red beetroot supplementation in health and disease. Nutrients 7:2801–2822. https://doi.org/10.3390/nu7042801
Muñoz-González C, Brule M, Martin C, Feron G, Canon F (2022) Molecular mechanisms of aroma persistence: from noncovalent interactions between aroma compounds and the oral mucosa to metabolization of aroma compounds by saliva and oral cells. Food Chem 373:131467. https://doi.org/10.1016/j.foodchem.2021.131467
Hou T, Sana SS, Li H, Xing Y, Nanda A et al (2022) Essential oils and its antibacterial, antifungal and anti-oxidant activity applications: a review. Food Biosci 47:101716. https://doi.org/10.1016/j.fbio.2022.101716
Munda S, Dutta S, Pandey SK, Sarma N, Lal M (2019) Antimicrobial activity of essential oils of medicinal and aromatic plants of the North east India: a biodiversity hot spot. J Essent Oil-Bear Plants 22:105–119. https://doi.org/10.1080/0972060X.2019.1601032
Aguirre-Becerra H, Vazquez-Hernandez MC, de la Saenz OD, Alvarado-Mariana A, Guevara-Gonzalez RG et al (2021) Role of stress and defense in plant secondary metabolites production. In: Pal D, Nayak AK (eds) Bioactive natural products for pharmaceutical applications. Springer, Berlin, pp 151–195. https://doi.org/10.1007/978-3-030-54027-2-5
Ju J, Xie Y, Yu H, Guo Y, Cheng Y et al (2022) Synergistic interactions of plant essential oils with antimicrobial agents: a new antimicrobial therapy. Crit Rev Food Sci Nutr 62:1740–1751. https://doi.org/10.1080/10408398.2020.1846494
Sugier P, Jakubowicz-Gil J, Sugier D, Kowalski R, Gawlik-Dziki U et al (2020) Chemical characteristics and anticancer activity of essential oil from Arnica montana L. rhizomes and roots. Molecules 25:1284. https://doi.org/10.3390/molecules25061284
Paes de Almeida V, Tolouei SEL, Minteguiaga M, Chaves DSDA, Heiden G et al (2023) Chemical profiles and cytotoxic activities of essential oils from six species of Baccharis subgenus Coridifoliae (Asteraceae). Chem Biodivers 20:e202300862. https://doi.org/10.1002/cbdv.202300862
Baptista RC, Horita CN, Sant’Ana AS (2020) Natural products with preservative properties for enhancing the microbiological safety and extending the shelf-life of seafood: a review. Food Res Int 127:108762. https://doi.org/10.1016/j.foodres.2019.108762
Al-Hwaiti MS, Alsbou EM, Abu Sheikha G, Bakchiche B, Pham TH et al (2021) Evaluation of the anticancer activity and fatty acids composition of “Handal” (Citrullus colocynthis L.) seed oil, a desert plant from south Jordan. Food Sci Nutr 9:282–289. https://doi.org/10.1002/fsn3.1994
Balusamy SR, Perumalsamy H, Veerappan K, Huq MA, Rajeshkumar S et al (2020) Citral induced apoptosis through modulation of key genes involved in fatty acid biosynthesis in human prostate cancer cells: In silico and in vitro study. BioMed Res Int 2020:6040727. https://doi.org/10.1155/2020/6040727
Kopustinskiene DM, Jakstas V, Savickas A, Bernatoniene J (2020) Flavonoids as anticancer agents. Nutrients 12:457. https://doi.org/10.3390/nu12020457
El-Amier YA, Soliman HM, El-Halawany EF, El-Nabawy BS (2022) Chemical characterization of Reichardia tingitana methanolic extract and evaluation of its antioxidant and anticancer activity. Egypt J Chem 65:933–940. https://doi.org/10.21608/EJCHEM20221557206722
Morsi EA, Ahmed HO, Abdel-Hady H, El-Sayed M, Shemis MA (2020) GC-analysis, and antioxidant, anti-inflammatory, and anticancer activities of some extracts and fractions of Linum usitatissimum. Curr Bioact Compd 16:1306–1318. https://doi.org/10.2174/1573407216666200206095954
Linz MS, Mattappallil A, Finkel D, Parker D (2023) Clinical impact of Staphylococcus aureus skin and soft tissue infections. Antibiotics 12:557. https://doi.org/10.3390/antibiotics12030557
Hurst JR, Shannon BA, Craig HC, Rishi A, Tuffs SW et al (2022) The Streptococcus pyogenes hyaluronic acid capsule promotes experimental nasal and skin infection by preventing neutrophil-mediated clearance. PLoS Pathog 18:e1011013. https://doi.org/10.1371/journal.ppat.1011013
Alamiri F, André O, De S, Nordenfelt P, Hakansson AP (2023) Role of serotype and virulence determinants of Streptococcus pyogenes biofilm bacteria in internalization and persistence in epithelial cells in vitro. Front Cell Infect Microbiol 13:1146431. https://doi.org/10.3389/fcimb.2023.1146431
Eishi Y (2023) Potential association of Cutibacterium acnes with sarcoidosis as an endogenous hypersensitivity infection. Microorganisms 11:289. https://doi.org/10.3390/microorganisms11020289
Mastrolorenzo A, Giomi B, Cipollini EM, Tammaro R, Decarli N et al (2012) Mycetomatoid infection of the penis by Candida albicans. Int J Dermatol 51:1082–1085. https://doi.org/10.1111/j.1365-4632.2011.05386.x
Jeffery-Smith A, Taori SK, Schelenz S, Jeffery K, Johnson EM et al (2018) Candida auris: a review of the literature. Clin Microbiol Rev 31:e00029-e117. https://doi.org/10.1128/cmr.00029-17
Perlin DS, Shor E, Zhao Y (2015) Update on antifungal drug resistance. Curr Clin Microbiol Rep 2:84–95. https://doi.org/10.1007/s40588-015-0015-1
Mustafa A, Turner C (2011) Pressurized liquid extraction as a green approach in food and herbal plants extraction: a review. Anal Chim Acta 703:8–18. https://doi.org/10.1016/j.aca.2011.07.018
Neto JAR, Alves SN, dos Santos Lima LAR (2023) Fatty acid methyl esters (FAMEs) obtained from edible vegetable oils: larvicidal activity and melanization process in Aedes aegypti larvae. Biocatal Agric Biotechnol 50:102689. https://doi.org/10.1016/j.bcab.2023.102689
Asraoui F, Kounnoun A, Cadi HE, Cacciola F et al (2021) Phytochemical investigation and antioxidant activity of Globularia alypum L Molecules 26:759. https://doi.org/10.3390/molecules26030759
Ben Mrid R, Bouchmaa N, Bouargalne Y, Ramdan B, Karrouchi K et al (2019) Phytochemical characterization, antioxidant and in vitro cytotoxic activity evaluation of Juniperus oxycedrus Subsp. oxycedrus needles and berries. Molecules 24:502. https://doi.org/10.3390/molecules24030502
Matuschek E, Copsey-Mawer S, Petersson S, Åhman J, Morris TE et al (2023) The European committee on antimicrobial susceptibility testing disc diffusion susceptibility testing method for frequently isolated anaerobic bacteria. Clin Microbiol Infect 29:795-e1. https://doi.org/10.1016/j.cmi.2023.01.027
Pesingi PV, Singh BR, Pesingi PK, Bhardwaj M, Singh SV et al (2019) MexAB-OprM efflux pump of Pseudomonas aeruginosa offers resistance to carvacrol: a herbal antimicrobial agent. Front Microbiol 10:2664. https://doi.org/10.3389/fmicb.2019.02664
Awang N, Aziz ZA, Kamaludin NF, Chan KM (2014) Cytotoxicity and mode of cell death induced by Triphenyltin (IV) compounds in vitro. OnLine J Bio Sci 14:84. https://doi.org/10.3844/ojbssp.2014.84.93
Maciejewski R, Radzikowska-Büchner E, Flieger W, Kulczycka K, Baj J et al (2022) An overview of essential microelements and common metallic nanoparticles and their effects on male fertility. Int J Environ Res Public Health 19:11066. https://doi.org/10.3390/ijerph191711066
Özturan K, Atasever M (2018) Süt ve ürünlerinde mineral maddeler ve ağır metaller. Atatürk Univ J Vet Sci 13:229–2412. https://doi.org/10.17094/ataunivbd.317822
El-Jendoubi H, Vázquez S, Calatayud A, Vavpetic P, Vogel-Mikuš K et al (2014) The effects of foliar fertilization with iron sulfate in chlorotic leaves are limited to the treated area. A study with peach trees (Prunus persica L. Batsch) grown in the field and sugar beet (Beta vulgaris L.) grown in hydroponics. Front Plant Sci 5:16. https://doi.org/10.3389/fpls.2014.00002
Mzoughi Z, Chahdoura H, Chakroun Y, Camara M, Fernandez-Ruiz V et al (2019) Wild edible Swiss chard leaves (Beta vulgaris L. var. cicla): Nutritional, phytochemical composition and biological activities. Food Res Int 119:612–621. https://doi.org/10.1016/j.foodres.2018.10.039
Biondo P, Boeing JS, Barizao EO, Souza N, Matsushita M et al (2014) Evaluation of beetroot (Beta vulgaris L.) leaves during its developmental stages: a chemical composition study. Food Sci Technol 34:94–101. https://doi.org/10.1590/S0101-20612014005000007
Zardi-Bergaoui A, BenNejma A, Harzallah-Skhiri F, Flamini G, Ascrizzi R et al (2017) Chemical composition and biological studies of the essential oil from aerial parts of Beta vulgaris subsp. maritima (L.) arcang. growing in Tunisia. Chem Biodivers 14:e1700234. https://doi.org/10.1002/cbdv.201700234
Onanuga AO, Okpala EO (2022) Chemical compositions and antioxidant activity of volatile oils from Morinda citrifolia and Beta vulgaris leaves from Nigeria. Biol Med Nat Prod Chem 11:161–167. https://doi.org/10.14421/biomedich.2022.112.161-167
Jaouadi R, Boussaid M, Zaouali Y (2023) Variation in essential oil composition within and among Tunisian Thymus algeriensis Boiss et Reut. (Lamiaceae) populations: effect of ecological factors and incidence on antiacetylcholinesterase and antioxidant activities. Biochem Syst Ecol 106:104543. https://doi.org/10.1016/j.bse.2022.104543
Ninfali P, Angelino D (2013) Nutritional and functional potential of Beta vulgaris cicla and rubra. Fitoterapia 89:188–199. https://doi.org/10.1016/j.fitote.2013.06.004
Kazemi Shahandashti SS, Maali Amiri R, Zeinali H, Ramezanpour SS (2013) Change in membrane fatty acid compositions and cold-induced responses in chickpea. Mol Biol Rep 40:893–903. https://doi.org/10.1007/s11033-012-2130-x
Salachas G, Giannakopoulos E, Hela D, Papasavvas A, Savvas D et al (2022) Enhancing bioactive compounds accumulation in red beet (Beta vulgaris L.) plants by managing N nutrition. The identification of the ‘critical’ zone as a cultivation technique. Plant Physiol Biochem 188:21–30. https://doi.org/10.1016/j.plaphy.2022.08.003
Gamba M, Raguindin PF, Asllanaj E, Merlo F, Glisic M et al (2021) Bioactive compounds and nutritional composition of Swiss chard (Beta vulgaris L. var. cicla and flavescens): a systematic review. Crit Rev Food Sci Nutr 61:3465–3480. https://doi.org/10.1080/10408398.2020.1799326
Georgiev VG, Weber J, Kneschke EM, Denev PN, Bley T et al (2010) Antioxidant activity and phenolic content of betalain extracts from intact plants and hairy root cultures of the red beetroot Beta vulgaris cv. Detroit dark red. Plant Foods Hum Nutr 65:105–111. https://doi.org/10.1007/s11130-010-0156-6
Gennari L, Felletti M, Blasa M, Angelino D, Celeghini C et al (2011) Total extract of Beta vulgaris var. Cicla seeds versus its purified phenolic components: antioxidant activities and antiproliferative effects against colon cancer cells. Phytochem Anal 22:272–279. https://doi.org/10.1002/pca.1276
Farhadi N, Babaei K, Farsaraei S, Moghaddam M, Pirbalouti AG (2020) Changes in essential oil compositions, total phenol, flavonoids and antioxidant capacity of Achillea millefolium at different growth stages. Ind Crops Prod 152:112570. https://doi.org/10.1016/j.indcrop.2020.112570
Ahmed SH (2023) Allium Cepa and Beta vulgaris extracts and their synergistic activity with antifungal against Candida albicans. South Asian Res J Pharm Sci 5:53–59. https://doi.org/10.36346/sarjps.2023.v05i03.003
Akbari B, Baghaei-Yazdi N, Bahmaie M, Mahdavi Abhari F (2022) The role of plant-derived natural antioxidants in reduction of oxidative stress. BioFactors 48:611–633. https://doi.org/10.1002/biof.1831
Rana A, Samtiya M, Dhewa T, Mishra V, Aluko RE (2022) Health benefits of polyphenols: a concise review. J Food Biochem 46:4264. https://doi.org/10.1111/jfbc.14264
Koochak H, Seyyednejad SM, Motamedi H (2010) Preliminary study on the antibacterial activity of some medicinal plants of Khuzestan (Iran). Asian Pac J Trop Med 3:180–184. https://doi.org/10.1016/S1995-7645(10)60004-1
Vulić JJ, Ćebović TN, Čanadanović-Brunet JM, Ćetković GS, Čanadanović VM et al (2014) In vivo and in vitro antioxidant effects of beetroot pomace extracts. J Funct Foods 6:168–175. https://doi.org/10.1016/j.jff.2013.10.003
John S, Monica J, Priyadarshini S, Sivaraj C, Arumugam P (2017) Antioxidant and antibacterial activities of Beta vulgaris l. peel extracts. Int J Pharma Res Health Sci 5:1974–79. https://doi.org/10.21276/ijprhs.2017.06.14
Verma S, Chaudhary HS (2011) Effect of Carissa carandas against clinically pathogenic bacterial strains. J Pharm Res 4:3769–3771
El-Zahar KM, Al-Jamaan ME, Al-Mutairi FR, Al-Hudiab AM, Al-Einzi MS et al (2022) Antioxidant, antibacterial, and antifungal activities of the ethanolic extract obtained from Berberis vulgaris roots and leaves. Molecules 27:6114. https://doi.org/10.3390/molecules27186114
Kamli MR, Srivastava V, Hajrah NH, Sabir JS, Ali A et al (2021) Phytogenic fabrication of Ag–Fe bimetallic nanoparticles for cell cycle arrest and apoptosis signaling pathways in Candida auris by generating oxidative stress. Antioxidants 10:182. https://doi.org/10.3390/antiox10020182
Mittal S, Roy S, Srivastava JN (2013) Fungicidal response of a novel natural photosensitizer (Beta vulgaris) on Candida albicans with low-power laser radiation. Laser Phys 23:055606. https://doi.org/10.1088/1054-660X/23/5/055606
Saha M, Bandyopadhyay PK (2020) In vivo and in vitro antimicrobial activity of phytol, a diterpene molecule, isolated and characterized from Adhatoda vasica Nees. (Acanthaceae), to control severe bacterial disease of ornamental fish, Carassius auratus, caused by Bacillus licheniformis PKBMS16. Microb Pathog 141:103977. https://doi.org/10.1016/j.micpath.2020.103977
Yang PF, Lu H, Wang QB, Zhao ZW, Liu Q et al (2020) Chemical composition and antimicrobial activities of the essential oil from the leaves of Pterocephalus hookeri. Nat Prod Commun 15:1–5. https://doi.org/10.1177/1934578X20981239
Guimarães A, Venâncio A (2022) The potential of fatty acids and their derivatives as antifungal agents: a review. Toxins 14:188. https://doi.org/10.3390/toxins14030188
https://www.who.int/docs/default-source/antimicrobial-resistance/amr-factsheet.pdf. Accessed 15.02.2024.
Alastruey-Izquierdo A (2022) WHO fungal priority pathogens list to guide research, development and public health action. World Health Organization: Geneva, Switzerland. http://hdl.handle.net/20.500.12105/15113.
World Health Organization (2021) Global antimicrobial resistance and use surveillance system (GLASS) report: 2021.
Romero SA, Pavan I, Morelli AP, Mancini M, da Silva L et al (2021) Anticancer effects of root and beet leaf extracts (Beta vulgaris L.) in cervical cancer cells (HeLa). Phytother Res 35:6191–6203. https://doi.org/10.1002/ptr.7255
Bouchmaa N, Mrid RB, Kabach I, Zouaoui Z, Karrouchi K et al (2022) Beta vulgaris subsp. maritima: a valuable food with high added health benefits. Appl Sci 12:1866. https://doi.org/10.3390/app12041866
Scarpa ES, Giammanco M, Magnani M (2021) Gastrointestinal tumors: phytochemical and drug combinations targeting the hallmarks of cancer. Appl Sci 11:10077. https://doi.org/10.3390/app112110077
Dhama K, Sharun K, Gugjoo MB, Tiwari R, Alagawany M et al (2023) A comprehensive review on chemical profile and pharmacological activities of Ocimum basilicum. Food Rev Int 39:119–147. https://doi.org/10.1080/87559129.2021.1900230
Yang J, Pi C, Wang G (2018) Inhibition of PI3K/Akt/mTOR pathway by apigenin induces apoptosis and autophagy in hepatocellular carcinoma cells. Biomed Pharmacother 103:699–707. https://doi.org/10.1016/j.biopha.2018.04.072
Liu R, Ji P, Liu B, Qiao H, Wang X et al (2017) Apigenin enhances the cisplatin cytotoxic effect through p53-modulated apoptosis. Oncol Lett 13:1024–1030. https://doi.org/10.3892/ol.2016.5495
Hadipour E, Taleghani A, Tayarani-Najaran N, Tayarani-Najaran Z (2020) Biological effects of red beetroot and betalains: a review. Phytother Res 34:1847–1867. https://doi.org/10.1002/ptr.6653
Bouyahya A, Belmehdi O, Benjouad A, El Hassani RA, Amzazi S et al (2020) Pharmacological properties and mechanism insights of Moroccan anticancer medicinal plants: what are the next steps? Ind Crops Prod 147:112198. https://doi.org/10.1016/j.indcrop.2020.112198
Al-Sheddi ES, Al-Zaid NA, Al-Oqail MM, Al-Massarani SM, El-Gamal AA et al (2019) Evaluation of cytotoxicity, cell cycle arrest and apoptosis induced by Anethum graveolens L. essential oil in human hepatocellular carcinoma cell line. Saudi Pharm J 27:1053–1060. https://doi.org/10.1016/j.jsps.2019.09.001
Rajput S, Kumar BP, Dey KK, Pal I, Parekh A et al (2013) Molecular targeting of Akt by thymoquinone promotes G1 arrest through translation inhibition of cyclin D1 and induces apoptosis in breast cancer cells. Life Sci 93:783–790. https://doi.org/10.1016/j.lfs.2013.09.009
Andrade MA, Braga MA, Cesar PH, Trento MVC, Espósito MA et al (2018) Anticancer properties of essential oils: an overview. Curr Cancer Drug Targets 18:957–966. https://doi.org/10.2174/1568009618666180102105843
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of ınterest
The authors did not receive support from any organization for the submitted work. Author Alper ZÖNGÜR declares that they have no conflict of interest.
Ethical approval
Since the study did not involve clinical interaction with patients, informed consent was not required. All samples were commercially obtained.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zöngür, A. Antimicrobial, Antioxidant and Cytotoxic Effects of Essential Oil, Fatty Acids and Bioactive Compounds of Beta vulgaris var. crassa (Fodder Beet). Indian J Microbiol (2024). https://doi.org/10.1007/s12088-024-01269-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12088-024-01269-8