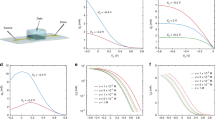
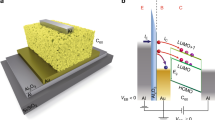
Abstract
Understanding the factors underpinning device switching times is crucial for the implementation of organic electrochemical transistors in neuromorphic computing, bioelectronics and real-time sensing applications. Existing models of device operation cannot explain the experimental observations that turn-off times are generally much faster than turn-on times in accumulation mode organic electrochemical transistors. Here, using operando optical microscopy, we image the local doping level of the transistor channel and show that turn-on occurs in two stages—propagation of a doping front, followed by uniform doping—while turn-off occurs in one stage. We attribute the faster turn-off to a combination of engineering as well as physical and chemical factors including channel geometry, differences in doping and dedoping kinetics and the phenomena of carrier-density-dependent mobility. We show that ion transport limits the operation speed in our devices. Our study provides insights into the kinetics of organic electrochemical transistors and guidelines for engineering faster organic electrochemical transistors.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the paper and its Supplementary Information. Source data are provided with this paper. Additional data may be requested from the authors.
References
Guo, K. et al. Rapid single-molecule detection of COVID-19 and MERS antigens via nanobody-functionalized organic electrochemical transistors. Nat. Biomed. Eng. 5, 666–677 (2021).
Bischak, C. G., Flagg, L. Q. & Ginger, D. S. Ion exchange gels allow organic electrochemical transistor operation with hydrophobic polymers in aqueous solution. Adv. Mater. 32, 2002610 (2020).
Strakosas, X., Bongo, M. & Owens, R. M. The organic electrochemical transistor for biological applications. J. Appl. Polym. Sci. 132, 41735 (2015).
Pappa, A. M. et al. Organic transistor arrays integrated with finger-powered microfluidics for multianalyte saliva testing. Adv. Healthc. Mater. 5, 2295–2302 (2016).
Huang, W. et al. Vertical organic electrochemical transistors for complementary circuits. Nature 613, 496–502 (2023).
Andersson Ersman, P. et al. All-printed large-scale integrated circuits based on organic electrochemical transistors. Nat. Commun. 10, 5053 (2019).
Fuller, E. J. et al. Parallel programming of an ionic floating-gate memory array for scalable neuromorphic computing. Science 364, 570–574 (2019).
Harikesh, P. C. et al. Ion-tunable antiambipolarity in mixed ion–electron conducting polymers enables biorealistic organic electrochemical neurons. Nat. Mater. 22, 242–248 (2023).
Chen, S. E., Giridharagopal, R. & Ginger, D. S. Artificial neuron transmits chemical signals. Nat. Mater. 22, 416–418 (2023).
Sarkar, T. et al. An organic artificial spiking neuron for in situ neuromorphic sensing and biointerfacing. Nat. Electron. 5, 774–783 (2022).
Yamamoto, S. & Malliaras, G. G. Controlling the neuromorphic behavior of organic electrochemical transistors by blending mixed and ion conductors. ACS Appl. Electron. Mater. 2, 2224–2228 (2020).
Gkoupidenis, P., Schaefer, N., Garlan, B. & Malliaras, G. G. Neuromorphic functions in PEDOT:PSS organic electrochemical transistors. Adv. Mater. 27, 7176–7180 (2015).
Khodagholy, D. et al. High transconductance organic electrochemical transistors. Nat. Commun. 4, 2133 (2013).
Lin, P., Yan, F. & Chan, H. L. W. Ion-sensitive properties of organic electrochemical transistors. Appl. Mater. Interfaces 2, 1637–1641 (2010).
Ghittorelli, M. et al. High-sensitivity ion detection at low voltages with current-driven organic electrochemical transistors. Nat. Commun. 9, 1441 (2018).
Gualandi, I. et al. Selective detection of dopamine with an all PEDOT:PSS organic electrochemical transistor. Sci. Rep. 6, 35419 (2016).
Xie, K. et al. Organic electrochemical transistor arrays for real-time mapping of evoked neurotransmitter release in vivo. eLife 9, e50345 (2020).
Khodagholy, D. et al. In vivo recordings of brain activity using organic transistors. Nat. Commun. 4, 1575 (2013).
Tang, X., Shen, H., Zhao, S., Li, N. & Liu, J. Flexible brain–computer interfaces. Nat. Electron. 6, 109–118 (2023).
Berggren, M., Głowacki, E. D., Simon, D. T., Stavrinidou, E. & Tybrandt, K. In vivo organic bioelectronics for neuromodulation. Chem. Rev. 122, 4826–4846 (2022).
Go, G.-T., Lee, Y., Seo, D.-G. & Lee, T.-W. Organic neuroelectronics: from neural interfaces to neuroprosthetics. Adv. Mater. 34, 2201864 (2022).
Van DeBurgt, Y., Melianas, A., Keene, S. T., Malliaras, G. & Salleo, A. Organic electronics for neuromorphic computing. Nat. Electron. 1, 386–397 (2018).
Gumyusenge, A., Melianas, A., Keene, S. T. & Salleo, A. Materials strategies for organic neuromorphic devices. Annu. Rev. Mater. Res. 51, 47–71 (2021).
Rivnay, J. et al. Organic electrochemical transistors. Nat. Rev. Mater. 3, 17086 (2018).
Zeglio, E. & Inganäs, O. Active materials for organic electrochemical transistors. Adv. Mater. 30, 1800941 (2018).
Kukhta, N. A., Marks, A. & Luscombe, C. K. Molecular design strategies toward improvement of charge injection and ionic conduction in organic mixed ionic–electronic conductors for organic electrochemical transistors. Chem. Rev. 122, 4325–4355 (2022).
He, Y., Kukhta, N. A., Marks, A. & Luscombe, C. K. The effect of side chain engineering on conjugated polymers in organic electrochemical transistors for bioelectronic applications. J. Mater. Chem. C 10, 2314–2332 (2022).
Paulsen, B. D., Tybrandt, K., Stavrinidou, E. & Rivnay, J. Organic mixed ionic–electronic conductors. Nat. Mater. 19, 13–26 (2020).
Flagg, L. Q. et al. P-Type electrochemical doping can occur by cation expulsion in a high-performing polymer for organic electrochemical transistors. ACS Mater. Lett. 2, 254–260 (2020).
Bernards, D. A. & Malliaras, G. G. Steady-state and transient behavior of organic electrochemical transistors. Adv. Funct. Mater. 17, 3538–3544 (2007).
Ohayon, D., Druet, V. & Inal, S. A guide for the characterization of organic electrochemical transistors and channel materials. Chem. Soc. Rev. 52, 1001–1023 (2023).
Inal, S., Malliaras, G. G. & Rivnay, J. Benchmarking organic mixed conductors for transistors. Nat. Commun. 8, 1767 (2017).
Li, P. & Lei, T. Molecular design strategies for high-performance organic electrochemical transistors. J. Polym. Sci. 60, 377–392 (2022).
Gentile, F. et al. A theoretical model for the time varying current in organic electrochemical transistors in a dynamic regime. Org. Electron. 35, 59–64 (2016).
Faria, G. C., Duong, D. T. & Salleo, A. On the transient response of organic electrochemical transistors. Org. Electron. 45, 215–221 (2017).
Paudel, P. R. et al. The transient response of organic electrochemical transistors. Adv. Theory Simul. 5, 2100563 (2022).
Keene, S. T. et al. Hole-limited electrochemical doping in conjugated polymers. Nat. Mater. 22, 1121–1127 (2023).
Ersman, P. A. et al. Screen printed digital circuits based on vertical organic electrochemical transistors. Flex. Print. Electron. 2, 045008 (2017).
Butler, J. A. V. Studies in heterogeneous equilibria. Part II.—The kinetic interpretation of the nernst theory of electromotive force. Trans. Faraday Soc. 19, 729–733 (1924).
Butler, J. A. V. Studies in heterogeneous equilibria. Part III. A kinetic theory of reversible oxidation potentials at inert electrodes. Trans. Faraday Soc. 19, 734–739 (1924).
Erdey-Grúz, T. & Volmer, M. Zur theorie der wasserstoff überspannung. Z. Phys. Chem. 150A, 203–213 (1930).
Bischak, C. G. et al. A reversible structural phase transition by electrochemically-driven ion injection into a conjugated polymer. J. Am. Chem. Soc. 142, 7434–7442 (2020).
Flagg, L. Q. et al. Polymer crystallinity controls water uptake in glycol side-chain polymer organic electrochemical transistors. J. Am. Chem. Soc. 141, 4345–4354 (2019).
Chen, S. E. et al. Impact of varying side chain structure on organic electrochemical transistor performance: a series of oligoethylene glycol-substituted polythiophenes. J. Mater. Chem. A 10, 10738–10749 (2022).
Neusser, D. et al. High conductivities of disordered P3HT films by an electrochemical doping strategy. Chem. Mater. 32, 6003–6013 (2020).
Hornberger, L. S., Neusser, D., Malacrida, C., Kaake, L. G. & Ludwigs, S. How charge trapping affects the conductivity of electrochemically doped poly(3-hexylthiophene) films. Appl. Phys. Lett. 119, 163301 (2021).
Jackson, S. R., Kingsford, R. L., Collins, G. W. & Bischak, C. G. Crystallinity determines ion injection kinetics and local ion density in organic mixed conductors. Chem. Mater. 35, 5392–5400 (2023).
Friedlein, J. T., Shaheen, S. E., Malliaras, G. G. & McLeod, R. R. Optical measurements revealing nonuniform hole mobility in organic electrochemical transistors. Adv. Electron. Mater. 1, 1500189 (2015).
Tanase, C., Meijer, E. J., Blom, P. W. M. & DeLeeuw, D. M. Unification of the hole transport in polymeric field-effect transistors and light-emitting diodes. Phys. Rev. Lett. 9, 216601 (2003).
Bässler, H., Kroh, D., Schauer, F., Nádaždy, V. & Köhler, A. Mapping the density of states distribution of organic semiconductors by employing energy resolved–electrochemical impedance spectroscopy. Adv. Funct. Mater. 31, 2007738 (2021).
Flagg, L. Q., Giridharagopal, R., Guo, J. & Ginger, D. S. Anion-dependent doping and charge transport in organic electrochemical transistors. Chem. Mater. 30, 5380–5389 (2018).
Wu, R., Paulsen, B. D., Ma, Q. & Rivnay, J. Mass and charge transport kinetics in an organic mixed ionic–electronic conductor. Chem. Mater. 34, 9699–9710 (2022).
Colucci, R., Barbosa, H. F. D. P., Günther, F., Cavassin, P. & Faria, G. C. Recent advances in modeling organic electrochemical transistors. Flex. Print. Electron. 5, 013001 (2020).
Guo, J. et al. Hydration of a side-chain-free n-type semiconducting ladder polymer driven by electrochemical doping. J. Am. Chem. Soc. 145, 1866–1876 (2023).
West, S. M. et al. Phenazine-substituted poly(benzimidazobenzophenanthrolinedione): electronic structure, thin film morphology, electron transport, and mechanical properties of an n-type semiconducting ladder polymer. Macromolecules 56, 2081–2091 (2023).
Zhang, Z. et al. Modulate molecular interaction between hole extraction polymers and lead ions toward hysteresis-free and efficient perovskite solar cells. Adv. Mater. Interfaces 5, 1800090 (2018).
Acknowledgements
This paper is based on research supported primarily by the National Science Foundation, first under DMR-2003456 and then under DMR-2309577. K.Y., Z.S. and C.-Z.L. acknowledge support from the National Natural Science Foundation of China (22125901) for supporting the synthesis of the PB2T-TEG polymer. J.W.O. and C.K.L.’s contributions to P3MEEMT polymer synthesis are based in part on work supported by the National Science Foundation, DMREF-1922259. Part of this work (transistor fabrication) was conducted at the Washington Nanofabrication Facility/Molecular Analysis Facility, a National Nanotechnology Coordinated Infrastructure (NNCI) site at the University of Washington with partial support from the National Science Foundation via awards NNCI-1542101 and NNCI-2025489.
Author information
Authors and Affiliations
Contributions
J.G. and S.E.C. contributed equally to the work. J.G., S.E.C. and D.S.G. conceived the project, designed the experiments and discussed the results together. J.G. and S.E.C. performed the experiments and analysed the data. S.E.C. wrote the first draft and J.G. made the figures. R.G. performed the SPICE circuit modelling. C.G.B designed the preliminary microscope experiment. K.Y., Z.S. and C.-Z.L. provided the PB2T-TEG polymer. J.W.O. and C.K.L. provided the P3MEEMT polymer. J.G., S.E.C., R.G. and D.S.G. revised the paper with input from all the authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Materials thanks the anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–20, Notes 1–8 and Tables 1–5.
Supplementary Video 1
Doping video coupled with the OECT drain current.
Supplementary Video 2
Comparison of doping front propagation at various VD values.
Supplementary Video 3
Five-cycle switching of PB2T-TEG.
Supplementary Video 4
P3MEEMT switching.
Supplementary Video 5
The VD and VG applied at the same time during transistor turn-on.
Supplementary Video 6
Dedoping video coupled with the OECT drain current.
Source data
Source Data Fig. 1
Source data for Fig. 1a.
Source Data Fig. 2
Source data for Fig. 2c–h.
Source Data Fig. 3
Source data for Fig. 3b,d–h,j,k.
Source Data Fig. 4
Source data for Fig. 4a,b.
Source Data Fig. 5
Source data for Fig. 5a–e.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guo, J., Chen, S.E., Giridharagopal, R. et al. Understanding asymmetric switching times in accumulation mode organic electrochemical transistors. Nat. Mater. (2024). https://doi.org/10.1038/s41563-024-01875-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41563-024-01875-3