Abstract
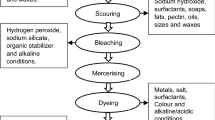
This study aims to solve the discharge of printing and dyeing reverse osmosis concentrate (PDROC) from a local printing and dyeing factory in Hangzhou, China. Electrochemical technique was used to treat the organic content of the concentrated wastewater. At the same time, the concentration levels of the target chemical species were double-checked using liquid–liquid extraction-gas chromatography–mass spectrometry and the UV–Vis spectroscopy. The findings revealed that the contaminants in the wastewater were primarily consisted of semi-volatile organic compounds such as long-chain alkanes, aromatics, and phthalates, with the concentration of dibutyl phthalate (DBP) being 64.4 µg/L. Decent removal efficiencies of chemical oxygen demand (COD) and DBP were observed after 3-h electro-degradation, which reached 73.6% and 45.8%, respectively. Additionally, the increment of current density and initial pH showed positive effects on the DBP degradation. Additionally, high current density and low starting pH are beneficial to DBP degradation. Furthermore, the by-products were analyzed by their MS spectrums to better understand the electro-degradation process of DBP, which primarily includes phthalic acid, ethane-1,1,2-tricarboxylic acid, hydroquinone, and oxalic acid, implying that the hydroxyl radicals (·OH) produced by electrolysis oxidized DBP in this solution via three proposed degradation pathways.






Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Aquino, J. M., Rocha-Filho, R. C., Ruotolo, L. A. M., Bocchi, N., & Biaggio, S. R. (2014). Electrochemical degradation of a real textile wastewater using β-PbO2 and DSA® anodes. Chemical Engineering Journal, 251, 138–145. https://doi.org/10.1016/j.cej.2014.04.032
Bagastyo, A. Y., Radjenovic, J., Mu, Y., Rozendal, R. A., Batstone, D. J., & Rabaey, K. (2011). Electrochemical oxidation of reverse osmosis concentrate on mixed metal oxide (MMO) titanium coated electrodes. WATER RESEARCH, 45(16), 4951–4959. https://doi.org/10.1016/j.watres.2011.06.039
Bagastyo, A. Y., Batstone, D. J., Kristiana, I., Gernjak, W., Joll, C., & Radjenouic, J. (2012). Electrochemical oxidation if reverse osmosis concentrate on boron-doped diamond anodes at circumneutral and acidic pH. Water Research, 46(18), 6104–6112. https://doi.org/10.1016/j.watres.2012.08.038
Bagastyo, A. Y., Batstone, D. J., Rabaey, K., & Radjenovic, J. (2013). Electrochemical oxidation of electrodialysed reverse osmosis concentrate on Ti/Pt–IrO2, Ti/SnO2–Sb and boron-doped diamond electrodes. Water Research, 47(1), 242–250. https://doi.org/10.1016/j.watres.2012.10.001
Boopathy, R., & Sekaran, G. (2014). Electrochemical treatment of reverse osmosis concentrate generated by the leather industry using a Cu-graphite electrode. RSC advances, 4(20), 9971–9979. https://doi.org/10.1039/c3ra45199b
Boopathy, R., Mandal, A. B., & Sekaran, G. (2014). Electrochemical treatment of evaporated residue of reverse osmosis concentrate generated from the leather industry. RSC Advances, 4(97), 54614–54624. https://doi.org/10.1039/c4ra09278c
Bu, F., Gao, B., Yue, Q., Shen, X., & Wang, W. (2019). Characterization of dissolved organic matter and membrane fouling in coagulation-ultrafiltration process treating micro-polluted surface water. Journal of Environmental Sciences, 75, 318–324. https://doi.org/10.1016/j.jes.2018.04.015
Chen, M., Pan, S., Zhang, C., Wang, C., Zhang, W., Chen, Z., Zhao, X., & Zhao, Y. (2020). Electrochemical oxidation of reverse osmosis concentrates using enhanced TiO2-NTA/SnO2-Sb anodes with/without PbO2 layer. Chemical Engineering Journal, 399, 125756. https://doi.org/10.1016/j.cej.2020.125756
Chen, M., Zhao, X., Wang, C., Pan, S., Zhang, C., & Wang, Y. (2021). Electrochemical oxidation of reverse osmosis concentrates using macroporous Ti-ENTA/SnO2-Sb flow-through anode: Degradation performance, energy efficiency and toxicity assessment. Journal of Hazardous Materials, 401, 123295. https://doi.org/10.1016/j.jhazmat.2020.123295
Chen, Y., Liao, D., Lin, Y., Deng, T., Yin, F., Su, P., & Feng, D. (2022). Electrochemical degradation performance and mechanism of dibutyl phthalate with hydrophobic PbO2 electrode. Chemosphere, 288, 132638. https://doi.org/10.1016/j.chemosphere.2021.132638
Chi, J., Zhang, H., & Zhao, D. (2021a). Impact of microplastic addition on degradation of dibutyl phthalate in offshore sediments. Marine Pollution Bulletin, 162, 111881. https://doi.org/10.1016/j.marpolbul.2020.111881
Chi, J., Zhang, H.T. & Zhao, D.X. (2021b). Impact of microplastic addition on degradation of dibutyl phthalate in offshore sediments. Marine Pollution Bulletin, 162.https://doi.org/10.1016/j.marpolbul.2020.111881
de Souza, F., Saez, C., Canizares, P., de Motheo, A., & Rodrigo, M. (2014a). Electrochemical removal of dimethyl phthalate with diamond anodes. Journal of Chemical Technology and Biotechnology, 89(2), 282–289. https://doi.org/10.1002/jctb.4118
Deng, D., Wu, X., Li, M., Qian, S., Tang, B., Wei, S., & Zhang, J. (2020). Electrochemical degradation of three phthalate esters in synthetic wastewater by using a Ce-doped Ti/PbO2 electrode. Chemosphere, 259, 127488. https://doi.org/10.1016/j.chemosphere.2020.127488
Fernandes, A., Labiadh, L., Ciriaco, L., Pacheco, M. J., Gadri, A., Ammar, S., & Lopes, A. (2017). Electro-Fenton oxidation of reverse osmosis concentrate from sanitary landfill leachate: Evaluation of operational parameters. Chemosphere, 184, 1223–1229. https://doi.org/10.1016/j.chemosphere.2017.06.088
Gao, B.-Y., Yue, Q.-Y., Wang, Y., & Zhou, W.-Z. (2007). Color removal from dye-containing wastewater by magnesium chloride. Journal of Environmental Management, 82(2), 167–172. https://doi.org/10.1016/j.jenvman.2005.12.019
He, L., Ji, Y., Cheng, J., Wang, C., Jiang, L., Chen, X., Li, H., Ke, S., & Wang, J. (2022). Effect of pH and Cl- concentration on the electrochemical oxidation of pyridine in low-salinity reverse osmosis concentrate: Kinetics, mechanism, and toxicity assessment. Chemical Engineering Journal, 449, 137669. https://doi.org/10.1016/j.cej.2022.137669
Hou, Y., Qu, J., Zhao, X., & Liu, H. (2009). Electrochemical incineration of dimethyl phthalate by anodic oxidation with boron-doped diamond electrode. Journal of Environmental Sciences, 21(10), 1321–1328. https://doi.org/10.1016/S1001-0742(08)62421-3
Huang, K., & MacKay, A. A. (2020). Microcystin-LR degradation kinetics during chlorination: Role of water quality conditions. Water Research, 185, 116305. https://doi.org/10.1016/j.watres.2020.116305
Hurwitz, G., Hoek, E. M. V., Liu, K., Fan, L., & Roddick, F. A. (2014). Photo-assisted electrochemical treatment of municipal wastewater reverse osmosis concentrate. Chemical Engineering Journal, 249, 180–188. https://doi.org/10.1016/j.cej.2014.03.084
Jamil, S., Loganathan, P., Kandasamy, J., Listowski, A., Khourshed, C., Naidu, R., & Vigneswaran, S. (2019). Removal of dissolved organic matter fractions from reverse osmosis concentrate: Comparing granular activated carbon and ion exchange resin adsorbents. Journal of Environmental Chemical Engineering, 7(3), 103126. https://doi.org/10.1016/j.jece.2019.103126
Jia, S., Han, Y., Zhuang, H., Han, H., & Li, K. (2017). Simultaneous removal of organic matter and salt ions from coal gasification wastewater RO concentrate and microorganisms succession in a MBR. Bioresource Technology, 241, 517–524. https://doi.org/10.1016/j.biortech.2017.05.158
Jiang, J., Li, W., Zhang, X., Liu, J., & Zhu, X. (2018). A new approach to controlling halogenated DBPs by GAC adsorption of aromatic intermediates from chlorine disinfection: Effects of bromide and contact time. Separation and Purification Technology, 203, 260–267. https://doi.org/10.1016/j.seppur.2018.04.050
Jiao, Y., Liu, L., Zhang, Q., Zhou, M., & Zhang, Y. (2021). Treatment of reverse osmosis concentrate from industrial coal wastewater using an electro-peroxone process with a natural air diffusion electrode. Separation and Purification Technology, 279, 119667. https://doi.org/10.1016/j.seppur.2021.119667
Labiadh, L., Fernandes, A., Ciríaco, L., Pacheco, M. J., Gadri, A., Ammar, S., & Lopes, A. (2016). Electrochemical treatment of concentrate from reverse osmosis of sanitary landfill leachate. Journal of Environmental Management, 181, 515–521. https://doi.org/10.1016/j.jenvman.2016.06.069
Li, H., Zhu, X., Jiang, Y., & Ni, J. (2010). Comparative electrochemical degradation of phthalic acid esters using boron-doped diamond and Pt anodes. Chemosphere, 80(8), 845–851. https://doi.org/10.1016/j.chemosphere.2010.06.006
Liao, D., Chen, Y., Yin, F., Lv, B., Wu, F., Xie, J., & Feng, D. (2023). Performance of electrochemical treatment of refractory organic matter in printing and dyeing reverse osmosis concentrate. Journal of Environmental Chemical Engineering, 11(1), 109173. https://doi.org/10.1016/j.jece.2022.109173
Lin, L., Zhang, S., Dong, L., Cao, Y., Zhang, W., Pan, X., Li, Y., Zhang, C., Tao, J., Jia, D., & Crittenden, J. (2023). Photodegradation behavior and mechanism of dibutyl phthalate in water under flood discharge atomization. Science of the Total Environment, 871, 161822. https://doi.org/10.1016/j.scitotenv.2023.161822
Liu, X., & He, Z. (2020). Decreased formation of disinfection by-products during electrochemical leachate oxidation and their post-removal by electro-adsorption. Science of the Total Environment, 730, 139171. https://doi.org/10.1016/j.scitotenv.2020.139171
Maharaja, P., Boopathy, R., Karthikeyan, S., Mahesh, M., Komal, A. S., Gupta, V. K., & Sekaran, G. (2016). Advanced oxidation of catechol in reverse osmosis concentrate generated in leather wastewater by Cu-graphite electrode. International Journal of Environmental Science and Technology, 13(9), 2143–2152. https://doi.org/10.1007/s13762-016-1044-x
Malamis, S., Katsou, E., Takopoulos, K., Demetriou, P., & Loizidou, M. (2012). Assessment of metal removal, biomass activity and RO concentrate treatment in an MBR–RO system. Journal of Hazardous Materials, 209–210, 1–8. https://doi.org/10.1016/j.jhazmat.2011.10.085
Meng, X. Y., Chen, Z. F., Wang, C., Zhang, W. Q., Zhang, K. H., Zhou, S. Q., Luo, J. M., Liu, N. A., Zhou, D. D., Li, D., & Crittenden, J. (2019). Development of a three-dimensional electrochemical system using a blue TiO2/SnO2-Sb2O3 anode for treating low-ionic-strength wastewater. Environmental Science & Technology, 53(23), 13784–13793. https://doi.org/10.1021/acs.est.9b05488
Nozari, M., Malakootian, M., Fard, N. J. H., & Mahmoudi-Moghaddam, H. (2022). Synthesis of Fe3O4@PAC as a magnetic nano-composite for adsorption of dibutyl phthalate from the aqueous medium: Modeling, analysis and optimization using the response surface methodology. Surfaces and Interfaces, 31, 101981. https://doi.org/10.1016/j.surfin.2022.101981
Pan, K., Tian, M., Jiang, Z. H., Kjartanson, B., & Chen, A. C. (2012). Electrochemical oxidation of lignin at lead dioxide nanoparticles photoelectrodeposited on TiO2 nanotube arrays. Electrochimica Acta, 60, 147–153. https://doi.org/10.1016/j.electacta.2011.11.025
Sarathy, S.R., Stefan, M.I., Royce, A. & Mohseni, M. (2011). Pilot-scale UV/H2O2 advanced oxidation process for surface water treatment and downstream biological treatment: Effects on natural organic matter characteristics and DBP formation potential. Environmental Technology 32(15), 1709–1718. https://doi.org/10.1080/09593330.2011.553843.
Scialdone, O., Galia, A., & Randazzo, S. (2011). Oxidation of carboxylic acids in water at IrO2-Ta2O5 and boron doped diamond anodes. Chemical Engineering Journal, 174(1), 266–274. https://doi.org/10.1016/j.cej.2011.09.016
Souza, F. L., Aquino, J. M., Irikura, K., Miwa, D. W., Rodrigo, M. A., & Motheo, A. J. (2014b). Electrochemical degradation of the dimethyl phthalate ester on a fluoride-doped Ti/β-PbO2 anode. Chemosphere, 109, 187–194. https://doi.org/10.1016/j.chemosphere.2014.02.018
Souza, F. L., Aquino, J. M., Miwa, D. W., Rodrigo, M. A., & Motheo, A. J. (2014c). Photo-assisted electrochemical degradation of the dimethyl phthalate ester on DSA® electrode. Journal of Environmental Chemical Engineering, 2(2), 811–818. https://doi.org/10.1016/j.jece.2014.02.003
Sun, Y.-X., Gao, Y., Hu, H.-Y., Tang, F., & Yang, Z. (2014). Characterization and biotoxicity assessment of dissolved organic matter in RO concentrate from a municipal wastewater reclamation reverse osmosis system. Chemosphere, 117, 545–551. https://doi.org/10.1016/j.chemosphere.2014.09.024
Sun, J.F., Wei, B., Mei, Q., An, Z.X., Wang, X.Y., Han, D.D., Xie, J., Zhan, J.H., Zhang, Q.Z., Wang, W.X. & He, M.X. (2020). Theoretical investigation on the degradation of dibutyl phthalate initiated by (OH)-O-center dot and SO4 center dot- in aqueous solution: Mechanism, kinetics and ecotoxicity assessment. Chemical Engineering Journal, 382. https://doi.org/10.1016/j.cej.2019.122791.
Umar, M., Roddick, F. A., Fan, L., Autin, O., & Jefferson, B. (2015). Treatment of municipal wastewater reverse osmosis concentrate using UVC-LED/H2O2 with and without coagulation pre-treatment. Chemical Engineering Journal, 260, 649–656. https://doi.org/10.1016/j.cej.2014.09.028
Wang, J., & Wang, S. (2018). Activation of persulfate (PS) and peroxymonosulfate (PMS) and application for the degradation of emerging contaminants. Chemical Engineering Journal, 334, 1502–1517. https://doi.org/10.1016/j.cej.2017.11.059
Wang, D., Duan, X. D., He, X. X., & Dionysiou, D. D. (2016). Degradation of dibutyl phthalate (DBP) by UV-254 nm/H2O2 photochemical oxidation: Kinetics and influence of various process parameters. Environmental Science and Pollution Research, 23(23), 23772–23780. https://doi.org/10.1007/s11356-016-7569-1
Wang, J., Zhang, T., Mei, Y., & Pan, B. (2018). Treatment of reverse-osmosis concentrate of printing and dyeing wastewater by electro-oxidation process with controlled oxidation-reduction potential (ORP). Chemosphere, 201, 621–626. https://doi.org/10.1016/j.chemosphere.2018.03.051
Wang, G., Zhang, Q., Chen, Q., Ma, X., Xin, Y., Zhu, X., Ma, D., Cui, C., Zhang, J., & Xiao, Z. (2019). Photocatalytic degradation performance and mechanism of dibutyl phthalate by graphene/TiO2 nanotube array photoelectrodes. Chemical Engineering Journal, 358, 1083–1090. https://doi.org/10.1016/j.cej.2018.10.039
Wang, L., Fu, G.Y., Zhao, B., Zhang, Z.H., Guo, X.F. & Zhang, H.W. (2014). Degradation of di-n-butyl phthalate in aqueous solution by the O-3/UV process. Desalination and Water Treatment, 52(4–6), 824–833. https://doi.org/10.1080/19443994.2013.826844.
Wei, F., Liao, D., Lin, Y., Hu, C., Ju, J., Chen, Y., & Feng, D. (2021). Electrochemical degradation of reverse osmosis concentrate (ROC) using the electrodeposited Ti/TiO2-NTs/PbO2 electrode. Separation and Purification Technology, 258, 118056. https://doi.org/10.1016/j.seppur.2020.118056
Westerhoff, P., Moon, H., Minakata, D., & Crittenden, J. (2009). Oxidation of organics in retentates from reverse osmosis wastewater reuse facilities. Water Research, 43(16), 3992–3998. https://doi.org/10.1016/j.watres.2009.04.010
Xiang, Q., Nomura, Y., Fukahori, S., Mizuno, T., Tanaka, H., & Fujiwara, T. (2019). Innovative treatment of organic contaminants in reverse osmosis concentrate from water reuse: A mini review. Current Pollution Reports, 5(4), 294–307. https://doi.org/10.1007/s40726-019-00119-2
Yao, J. Y., Yu, Y., Qu, R. J., Chen, J., Huo, Z. L., Zhu, F., & Wang, Z. Y. (2020). Fe-activated peroxymonosulfate enhances the degradation of dibutyl phthalate on ground quartz sand. Environmental Science & Technology, 54(14), 9052–9061. https://doi.org/10.1021/acs.est.0c00793
Yin, X., Li, W., Zhu, H., Yu, J., Wei, K., Gao, Z., Zhang, Y., Chen, H., Gu, L., & Han, W. (2024). Electrochemical treatment of municipal reverse osmosis concentrates by a TiO2-BNTs/SnO2-Sb reactive electrochemical membrane. Separation and Purification Technology, 331, 125726. https://doi.org/10.1016/j.seppur.2023.125726
Yuan, B. L., Li, X. Z., & Graham, N. (2008). Aqueous oxidation of dimethyl phthalate in a Fe(VI)-TiO2-UV reaction system. Water Research, 42(6–7), 1413–1420. https://doi.org/10.1016/j.watres.2007.10.010
Zhao, C., Wu, Z., Lai, J., Liu, L., Li, H., & Wang, H. (2024). Efficient electrochemical oxidation of refractory organics in actual petrochemical reverse osmosis concentrates by Ti/SnO2-Sb mesh anode. Process Safety and Environmental Protection, 182, 1060–1071. https://doi.org/10.1016/j.psep.2023.12.067
Zheng, L., Wang, X., & Wang, X. (2015). Reuse of reverse osmosis concentrate in textile and dyeing industry by combined process of persulfate oxidation and lime-soda softening. Journal of Cleaner Production, 108, 525–533. https://doi.org/10.1016/j.jclepro.2015.09.027
Zhou, M., Liu, L., Jiao, Y., Wang, Q., & Tan, Q. (2011). Treatment of high-salinity reverse osmosis concentrate by electrochemical oxidation on BDD and DSA electrodes. Desalination, 277(1), 201–206. https://doi.org/10.1016/j.desal.2011.04.030
Funding
This work was supported by the National Key R&D Program of China (2021YFC3100100).
Author information
Authors and Affiliations
Contributions
Dexiang Liao: conceptualization, writing—review and editing, validation, project administration, resources. Fan Wu: methodology, investigation, writing—original draft, formal analysis. Tianyu Deng: visualization, methodology. Fang Yin: formal analysis, software. Penghao Su: software, data curation. Daolun Feng: supervision, validation.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liao, D., Wu, F., Deng, T. et al. Electro-degradation of Dibutyl Phthalate (DBP) in Printing and Dyeing Reverse Osmosis Concentrate. Water Air Soil Pollut 235, 270 (2024). https://doi.org/10.1007/s11270-024-07051-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-024-07051-2