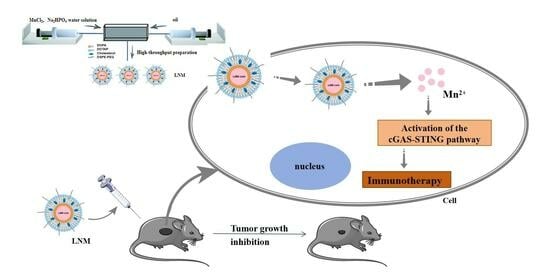
High Manganese Content of Lipid NanoMn (LNM) by Microfluidic Technology for Enhancing Anti-Tumor Immunity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Liposomes Using Microfluidics
2.3. LNM Activates cGAS-STING Pathway
2.4. Cytotoxicity Assay for LNM
2.5. Determination of Intracellular Manganese Content
2.6. Cell Uptake Was Detected Using Flow Cytometry
2.7. LNM Distribution Experiment
2.8. LNM Activates Immunity
2.9. Serum Cytokine Levels of Immunized Mice Were Detected by ELISA
2.10. Detection of Mouse Spleen Cells by Flow Cytometry
2.11. In Vivo Safety of LNM
2.12. Data Processing
3. Results
3.1. Process Screening of LNM Nanoparticles
3.1.1. The Effect of Flow Rate on the Particle Size of LNM Core
3.1.2. The Effect of the Ratio of Microprecipitates in the Inner Phospholipid on the Dispersion and Uniformity of the LNM Core
3.1.3. The Effect of the Ratio of the LNM Core to the Outer Phospholipid on the Load Ratio of Mn
3.2. The Characterization of LNM
3.3. The Verification of LNM Uptake and Immune Activation Ability
3.4. Anti-Tumor Effect of LNM Nanoparticles
3.5. Immunoactivation Effect of LNM
3.6. Safety Analysis of LNM
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yang, Z.; Gao, D.; Zhao, J.; Yang, G.; Guo, M.; Wang, Y.; Ren, X.; Kim, J.S.; Jin, L.; Tian, Z.; et al. Thermal immuno-nanomedicine in cancer. Nat. Rev. Clin. Oncol. 2023, 20, 116–134. [Google Scholar] [CrossRef]
- Sun, X.; Zhang, Y.; Li, J.; Park, K.S.; Han, K.; Zhou, X.; Xu, Y.; Nam, J.; Xu, J.; Shi, X.; et al. Amplifying STING activation by cyclic dinucleotide-manganese particles for local and systemic cancer metalloimmunotherapy. Nat. Nanotechnol. 2021, 16, 1260–1270. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Guan, Y.; Lv, M.; Zhang, R.; Guo, Z.; Wei, X.; Du, X.; Yang, J.; Li, T.; Wan, Y.; et al. Manganese Increases the Sensitivity of the cGAS-STING Pathway for Double-Stranded DNA and Is Required for the Host Defense against DNA Viruses. Immunity 2018, 48, 675–687.e7. [Google Scholar] [CrossRef]
- Zhao, Z.; Ma, Z.; Wang, B.; Guan, Y.; Su, X.D.; Jiang, Z. Mn2+ Directly Activates cGAS and Structural Analysis Suggests Mn2+ Induces a Noncanonical Catalytic Synthesis of 2′3′-cGAMP. Cell Rep. 2020, 32, 108053. [Google Scholar] [CrossRef] [PubMed]
- Lv, M.; Chen, M.; Zhang, R.; Zhang, W.; Wang, C.; Zhang, Y.; Wei, X.; Guan, Y.; Liu, J.; Feng, K.; et al. Manganese is critical for antitumor immune responses via cGAS-STING and improves the efficacy of clinical immunotherapy. Cell Res. 2020, 30, 966–979. [Google Scholar] [CrossRef] [PubMed]
- Zizzari, A.; Bianco, M.; Carbone, L.; Perrone, E.; Amato, F.; Maruccio, G.; Rendina, F.; Arima, V. Continuous-Flow Production of Injectable Liposomes via a Microfluidic Approach. Materials 2017, 10, 1411. [Google Scholar] [CrossRef] [PubMed]
- Maeki, M.; Uno, S.; Niwa, A.; Okada, Y.; Tokeshi, M. Microfluidic technologies and devices for lipid nanoparticle-based RNA delivery. J. Control. Release 2022, 344, 80–96. [Google Scholar] [CrossRef] [PubMed]
- Noviana, E.; Ozer, T.; Carrell, C.S.; Link, J.S.; McMahon, C.; Jang, I.; Henry, C.S. Microfluidic Paper-Based Analytical Devices: From Design to Applications. Chem. Rev. 2021, 121, 11835–11885. [Google Scholar] [CrossRef] [PubMed]
- Niculescu, A.G.; Chircov, C.; Bîrcă, A.C.; Grumezescu, A.M. Fabrication and Applications of Microfluidic Devices: A Review. Int. J. Mol. Sci. 2021, 22, 2011. [Google Scholar] [CrossRef]
- Li, Z.; Mak, S.Y.; Sauret, A.; Shum, H.C. Syringe-pump-induced fluctuation in all-aqueous microfluidic system implications for flow rate accuracy. Lab Chip 2014, 14, 744–749. [Google Scholar] [CrossRef]
- Zhao, W.; Hanson, L.; Lou, H.Y.; Akamatsu, M.; Chowdary, P.D.; Santoro, F.; Marks, J.R.; Grassart, A.; Drubin, D.G.; Cui, Y.; et al. Nanoscale manipulation of membrane curvature for probing endocytosis in live cells. Nat. Nanotechnol. 2017, 12, 750–756. [Google Scholar] [CrossRef] [PubMed]
- Holmfred, E.; Loeschner, K.; Sloth, J.J.; Jensen, K.A. Validation and Demonstration of an Atmosphere-Temperature-pH-Controlled Stirred Batch Reactor System for Determination of (Nano)Material Solubility and Dissolution Kinetics in Physiological Simulant Lung Fluids. Nanomaterials 2022, 12, 517. [Google Scholar] [CrossRef] [PubMed]
- Kirichenko, M.N.; Sanoeva, A.T.; Chaikov, L.L. Appearance of an artifact peak in the particle size distribution measured by DLS at low concentrations. Bull. Lebedev Phys. Inst. 2016, 43, 256–260. [Google Scholar] [CrossRef]
- Wang, P.; Rahman, M.A.; Zhao, Z.; Weiss, K.; Zhang, C.; Chen, Z.; Hurwitz, S.J.; Chen, Z.G.; Shin, D.M.; Ke, Y. Visualization of the Cellular Uptake and Trafficking of DNA Origami Nanostructures in Cancer Cells. J. Am. Chem. Soc. 2018, 140, 2478–2484. [Google Scholar] [CrossRef] [PubMed]
- Rennick, J.J.; Johnston, A.P.R.; Parton, R.G. Key principles and methods for studying the endocytosis of biological and nanoparticle therapeutics. Nat. Nanotechnol. 2021, 16, 266–276. [Google Scholar] [CrossRef]
- Lang, T.; Liu, Y.; Zheng, Z.; Ran, W.; Zhai, Y.; Yin, Q.; Zhang, P.; Li, Y. Cocktail Strategy Based on Spatio-Temporally Controlled Nano Device Improves Therapy of Breast Cancer. Adv. Mater. 2019, 31, e1806202. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.; Zhang, S.; Li, N.; Fan, P.; Hu, X.; Liang, K.; Cheng, X.; Wu, Y. Stable expression ratios of five pyroptosis-inducing cytokines in the spleen and thymus of mice showed potential immune regulation at the organ level. Lupus 2020, 29, 290–302. [Google Scholar] [CrossRef]
- Kim, H.I.; Kim, D.S.; Jung, Y.; Sung, N.Y.; Kim, M.; Han, I.J.; Nho, E.Y.; Hong, J.H.; Lee, J.K.; Boo, M.; et al. Immune-Enhancing Effect of Sargassum horneri on Cyclophosphamide-Induced Immunosuppression in BALB/c Mice and Primary Cultured Splenocytes. Molecules 2022, 27, 8253. [Google Scholar] [CrossRef]
- Zhao, L.; Liu, L.; Guo, B.; Zhu, B. Regulation of adaptive immune responses by guiding cell movements in the spleen. Front. Microbiol. 2015, 6, 645. [Google Scholar] [CrossRef]
- Hu, D.; Al-Shalan, H.A.M.; Shi, Z.; Wang, P.; Wu, Y.; Nicholls, P.K.; Greene, W.K.; Ma, B. Distribution of nerve fibers and nerve-immune cell association in mouse spleen revealed by immunofluorescent staining. Sci. Rep. 2020, 10, 9850. [Google Scholar] [CrossRef]
- Slezak, S.L.; Worschech, A.; Wang, E.; Stroncek, D.F.; Marincola, F.M. Analysis of vaccine-induced T cells in humans with cancer. Adv. Exp. Med. Biol. 2010, 684, 178–188. [Google Scholar] [CrossRef] [PubMed]
- Cachot, A.; Bilous, M.; Liu, Y.C.; Li, X.; Saillard, M.; Cenerenti, M.; Rockinger, G.A.; Wyss, T.; Guillaume, P.; Schmidt, J.; et al. Tumor-specific cytolytic CD4 T cells mediate immunity against human cancer. Sci. Adv. 2021, 7, eabe3348. [Google Scholar] [CrossRef] [PubMed]
- Raphael, I.; Joern, R.R.; Forsthuber, T.G. Memory CD4+ T Cells in Immunity and Autoimmune Diseases. Cells 2020, 9, 531. [Google Scholar] [CrossRef] [PubMed]
- Tanoue, T.; Morita, S.; Plichta, D.R.; Skelly, A.N.; Suda, W.; Sugiura, Y.; Narushima, S.; Vlamakis, H.; Motoo, I.; Sugita, K.; et al. A defined commensal consortium elicits CD8 T cells and anti-cancer immunity. Nature 2019, 565, 600–605. [Google Scholar] [CrossRef] [PubMed]
- Haubruck, P.; Colbath, A.C.; Liu, Y.; Stoner, S.; Shu, C.; Little, C.B. Flow Cytometry Analysis of Immune Cell Subsets within the Murine Spleen, Bone Marrow, Lymph Nodes and Synovial Tissue in an Osteoarthritis Model. J. Vis. Exp. 2020, 158, e61008. [Google Scholar] [CrossRef]
- Ma, D.; Zhu, X.; Zhao, P.; Zhao, C.; Li, X.; Zhu, Y.; Li, L.; Sun, J.; Peng, J.; Ji, C.; et al. Profile of Th17 cytokines (IL-17, TGF-beta, IL-6) and Th1 cytokine (IFN-gamma) in patients with immune thrombocytopenic purpura. Ann. Hematol. 2008, 87, 899–904. [Google Scholar] [CrossRef] [PubMed]
- Talaat, R.M.; Sibaii, H.; Bassyouni, I.H.; El-Wakkad, A. IL-17, IL-10, IL-6, and IFN-γ in Egyptian Behçet’s disease: Correlation with clinical manifestations. Eur. Cytokine Netw. 2019, 30, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Petruccioli, E.; Farroni, C.; Cuzzi, G.; Vanini, V.; Palmieri, F.; Vittozzi, P.; Goletti, D. VIDAS® TB-IGRA reagents induce a CD4+ and CD8+ T-cell IFN-γ response for both TB infection and active TB. Int. J. Tuberc. Lung Dis. 2022, 26, 65–68. [Google Scholar] [CrossRef] [PubMed]
- Sedighi, M.; Sieber, S.; Rahimi, F.; Shahbazi, M.A.; Rezayan, A.H.; Huwyler, J.; Witzigmann, D. Rapid optimization of liposome characteristics using a combined microfluidics and design-of-experiment approach. Drug Deliv. Transl. Res. 2019, 9, 404–413. [Google Scholar] [CrossRef]
- Ablasser, A.; Chen, Z.J. cGAS in action: Expanding roles in immunity and inflammation. Science 2019, 363, eaat8657. [Google Scholar] [CrossRef]
- Hooy, R.M.; Massaccesi, G.; Rousseau, K.E.; Chattergoon, M.A.; Sohn, J. Allosteric coupling between Mn2+ and dsDNA controls the catalytic efficiency and fidelity of cGAS. Nucleic Acids Res. 2020, 48, 4435–4447. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Ascano, M.; Wu, Y.; Barchet, W.; Gaffney, B.L.; Zillinger, T.; Serganov, A.A.; Liu, Y.; Jones, R.A.; Hartmann, G.; et al. Cyclic [G(2′,5′)pA(3′,5′)p] is the metazoan second messenger produced by DNA-activated cyclic GMP-AMP synthase. Cell 2013, 153, 1094–1107. [Google Scholar] [CrossRef] [PubMed]
- Smialowicz, R.J.; Riddle, M.M.; Rogers, R.R.; Luebke, R.W.; Burleson, G.R. Enhancement of natural killer cell activity and interferon production by manganese in young mice. Immunopharmacol. Immunotoxicol. 1988, 10, 93–107. [Google Scholar] [CrossRef] [PubMed]






| Sample | Flow Rate of MnCl2 (μL/min) | Flow Rate of Na2HPO4 (μL/min) | Flow Rate of Cyclohexane/CO-520 (mL/min) | FRR * | Particle Size (nm) |
|---|---|---|---|---|---|
| 1 | 42 | 42 | 2.1 | 0.02 | <10 nm |
| 2 | 100 | 100 | 2.1 | 0.048 | |
| 3 | 180 | 180 | 2.1 | 0.086 | |
| 4 | 210 | 210 | 2.1 | 0.1 | |
| 5 | 220 | 220 | 2.1 | 0.105 | |
| 6 | 250 | 250 | 2.1 | 0.119 | |
| 7 | 270 | 270 | 2.1 | 0.129 | 10–20 nm |
| 8 | 320 | 320 | 2.1 | 0.152 | |
| 9 | 350 | 350 | 2.1 | 0.167 | |
| 10 | 378 | 378 | 2.1 | 0.18 | |
| 11 | 420 | 420 | 2.1 | 0.2 | 30–50 nm |
| 12 | 438 | 438 | 2.1 | 0.209 | |
| 13 | 450 | 450 | 2.1 | 0.214 | |
| 14 | 470 | 470 | 2.1 | 0.223 | |
| 15 | 490 | 490 | 2.1 | 0.233 | |
| 16 | 520 | 520 | 2.1 | 0.248 | |
| 17 | 530 | 530 | 2.1 | 0.252 | |
| 18 | 550 | 550 | 2.1 | 0.262 | |
| 19 | 570 | 570 | 2.1 | 0.271 | >50 nm |
| 20 | 590 | 590 | 2.1 | 0.281 | |
| 21 | 600 | 600 | 2.1 | 0.286 | |
| 22 | 620 | 620 | 2.1 | 0.295 | |
| 23 | 640 | 640 | 2.1 | 0.305 | |
| 24 | 660 | 660 | 2.1 | 0.314 |
| Sample | DOPA Conc (mg/mL) | FRR | TFR * | Particle Size (nm) |
|---|---|---|---|---|
| 1 | 20 | 0.2 | 2520 | 30 |
| 2 | 23 | 0.2 | 2520 | 40 |
| 3 | 26 | 0.2 | 2520 | 20 |
| 4 | 30 | 0.2 | 2520 | 30–50 |
| 5 | 33 | 0.2 | 2520 | 30–50 |
| 6 | 36 | 0.2 | 2520 | 30–50 |
| Sample | Volume of LNM Core (μL) | Outer Lipid Content * (mM) | Particle Size (nm) | Mn2+ Content (105/mL) | Yield (%) |
|---|---|---|---|---|---|
| 1 | 100 | 2 | 44.9 | 11.6 | 14.06 |
| 2 | 100 | 5 | 53.3 | 12.7 | 15.39 |
| 3 | 100 | 10 | 53.9 | 34 | 19.7 |
| 4 | 200 | 5 | 56.5 | 43.8 | 16.45 |
| 5 | 200 | 10 | 55.19 | 48.4 | 26.5 |
| 6 | 300 | 10 | 53.79 | 48.8 | 29.3 |
| 7 | 400 | 10 | 45.8 | 54.3 | 39 |
| 8 | 500 | 10 | 45.2 | 160.9 | 41.2 |
| 9 | 600 | 10 | 56 | 239.7 | 58.11 |
| 10 | 1000 | 10 | 47.1 | 756.1 | 91.64 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, J.; Gong, J.; Gong, L.; Zhu, C.; Li-Yang, L.; Wang, J.; Yang, Y.; Zhang, S.; Liu, S.; Fu, J.-J.; et al. High Manganese Content of Lipid NanoMn (LNM) by Microfluidic Technology for Enhancing Anti-Tumor Immunity. Pharmaceutics 2024, 16, 556. https://doi.org/10.3390/pharmaceutics16040556
Sun J, Gong J, Gong L, Zhu C, Li-Yang L, Wang J, Yang Y, Zhang S, Liu S, Fu J-J, et al. High Manganese Content of Lipid NanoMn (LNM) by Microfluidic Technology for Enhancing Anti-Tumor Immunity. Pharmaceutics. 2024; 16(4):556. https://doi.org/10.3390/pharmaceutics16040556
Chicago/Turabian StyleSun, Jiawei, Jingjing Gong, Lidong Gong, Chuanda Zhu, Longhao Li-Yang, Jingya Wang, Yuanyuan Yang, Shiming Zhang, Silu Liu, Ji-Jun Fu, and et al. 2024. "High Manganese Content of Lipid NanoMn (LNM) by Microfluidic Technology for Enhancing Anti-Tumor Immunity" Pharmaceutics 16, no. 4: 556. https://doi.org/10.3390/pharmaceutics16040556