Abstract
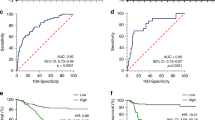
Gastric cancer (GC) is a serious malignant tumour with a high mortality rate and a poor prognosis. Recently, emerging evidence has suggested that N6-methyladenosine (m6A) modification plays a crucial regulatory role in cancer progression. However, the exact role of m6A regulatory factors FTO in GC is unclear. First, the expression of m6A methylation-related regulatory factors in clinical samples and the clinical data of the corresponding patients were obtained from The Cancer Genome Atlas (TCGA-STAD) dataset, and correlation analysis between FTO expression and patient clinicopathological parameters was subsequently performed. qRT-PCR, immunohistochemistry (IHC) and western blotting (WB) were used to verify FTO expression in GC. CCK-8, EdU, flow cytometry and transwell assays were used to evaluate the effect of FTO on the behaviour of GC cells. Transcriptome sequencing and RNA immunoprecipitation analysis were used to explore the potential regulatory mechanisms mediated by FTO. FTO was highly expressed in GC tissues and cells, and high expression of FTO predicted a worse prognosis than low expression. Functionally, overexpression of FTO promoted the proliferation, migration and invasion of GC cells but inhibited cell apoptosis. Mechanistically, we found that FTO is upregulated in GC and promotes GC progression by modulating the expression of MAP4K4. Taken together, our findings provide new insights into the effects of FTO-mediated m6A demethylation and could lead to the development of new strategies for GC monitoring and aggressive treatment.








Similar content being viewed by others
Data availability
Transcriptomic, matched clinical data and MAF files were downloaded from the TCGA database (https://portal.gdc.cancer.gov/).
References
Sung H, Ferlay J, Siegel R, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J Clin. 2021;71(3):209–49.
Zong L, Abe M, Seto Y, Ji J. The challenge of screening for early gastric cancer in China. Lancet. 2016;388(10060):2606.
Janjigian Y, Shitara K, Moehler M, Garrido M, Salman P, Shen L, Wyrwicz L, Yamaguchi K, Skoczylas T, Campos Bragagnoli A, et al. First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): a randomised, open-label, phase 3 trial. Lancet. 2021;398(10294):27–40.
Sun H, Li K, Liu C, Yi C. Regulation and functions of non-mA mRNA modifications. Nat Rev Mol Cell Biol. 2023;24(10):714–31.
Li D, Shen L, Zhang X, Chen Z, Huang P, Huang C, Qin S. LncRNA ELF3-AS1 inhibits gastric cancer by forming a negative feedback loop with SNAI2 and regulates ELF3 mRNA stability via interacting with ILF2/ILF3 complex. J Exp Clin Cancer Res. 2022;41(1):332.
Gerken T, Girard C, Tung Y, Webby C, Saudek V, Hewitson K, Yeo G, McDonough M, Cunliffe S, McNeill L, et al. The obesity-associated FTO gene encodes a 2-oxoglutarate-dependent nucleic acid demethylase. Science. 2007;318(5855):1469–72.
Li Y, Su R, Deng X, Chen Y, Chen J. FTO in cancer: functions, molecular mechanisms, and therapeutic implications. Trends Cancer. 2022;8(7):598–614.
Yang S, Wei J, Cui Y, Park G, Shah P, Deng Y, Aplin A, Lu Z, Hwang S, He C, et al. mA mRNA demethylase FTO regulates melanoma tumorigenicity and response to anti-PD-1 blockade. Nat Commun. 2019;10(1):2782.
Tang Z, Kang B, Li C, Chen T, Zhang Z. GEPIA2: an enhanced web server for large-scale expression profiling and interactive analysis. Nucleic Acids Res. 2019;47:W556–60.
Posta M, Győrffy B. Analysis of a large cohort of pancreatic cancer transcriptomic profiles to reveal the strongest prognostic factors. Clin Transl Sci. 2023;16(8):1479–91.
Huang H, Han Q, Zheng H, Wang J. MAP4K4 mediates the SOX6-induced autophagy and reduces the chemosensitivity of cervical cancer. Cell Death Dis. 2022. https://doi.org/10.1038/s41419-021-04474-1.
Juin A, Spence H, Machesky L. Dichotomous role of the serine/threonine kinase MAP4K4 in pancreatic ductal adenocarcinoma onset and metastasis through control of AKT and ERK pathways. J Pathol. 2024. https://doi.org/10.1002/path.6248.
Li L, Li Z, Meng X, Wang X, Song D, Liu Y, Xu T, Qin J, Sun N, Tian K, et al. Histone lactylation-derived LINC01127 promotes the self-renewal of glioblastoma stem cells via the cis-regulating the MAP4K4 to activate JNK pathway. Cancer Lett. 2023;579: 216467.
Chen K, Yuan X, Wang S, Zheng F, Fu Z, Shen Z, Cheng X, Wang Y, Tang S, Ni H, et al. MAP4K4 promotes ovarian cancer metastasis through diminishing ADAM10-dependent N-cadherin cleavage. Oncogene. 2023;42(18):1438–52.
Ding L, Jiang L, Xing Z, Dai H, Wei J. Map4k4 is up-regulated and modulates granulosa cell injury and oxidative stress in polycystic ovary syndrome via activating JNK/c-JUN pathway: an experimental study. Int Immunopharmacol. 2023;124: 110841.
Wan C, Chen W, Cui Y, He Z. MAP4K4/JNK signaling pathway stimulates proliferation and suppresses apoptosis of human spermatogonial stem cells and lower level of MAP4K4 is associated with male infertility. Cells. 2022. https://doi.org/10.3390/cells11233807.
Siegel R, Giaquinto A, Jemal A. Cancer statistics. CA. 2024;74(1):12–49.
Xu J, Jiang H, Pan Y, Gu K, Cang S, Han L, Shu Y, Li J, Zhao J, Pan H, et al. Sintilimab plus chemotherapy for unresectable gastric or gastroesophageal junction cancer: the ORIENT-16 randomized clinical trial. JAMA. 2023;330(21):2064–74.
Kelly R, Ajani J, Kuzdzal J, Zander T, Van Cutsem E, Piessen G, Mendez G, Feliciano J, Motoyama S, Lièvre A, et al. Adjuvant nivolumab in resected esophageal or gastroesophageal junction cancer. N Engl J Med. 2021;384(13):1191–203.
Li D, Xia L, Huang P, Wang Z, Guo Q, Huang C, Leng W, Qin S. Heterogeneity and plasticity of epithelial-mesenchymal transition (EMT) in cancer metastasis: focusing on partial EMT and regulatory mechanisms. Cell Prolif. 2023;56(6): e13423.
Liabeuf D, Oshima M, Stange D, Sigal M. Stem cells, Helicobacter pylori, and mutational landscape: utility of preclinical models to understand carcinogenesis and to direct management of gastric cancer. Gastroenterology. 2022;162(4):1067–87.
Grady W, Yu M, Markowitz S. Epigenetic alterations in the gastrointestinal tract: current and emerging use for biomarkers of cancer. Gastroenterology. 2021;160(3):690–709.
Li D, Xia L, Huang P, Wang Z, Guo Q, Huang C, Leng W, Qin S. Cancer-associated fibroblast-secreted IGFBP7 promotes gastric cancer by enhancing tumor associated macrophage infiltration via FGF2/FGFR1/PI3K/AKT axis. Cell Death Discov. 2023;9(1):17.
Lin J, Zhan G, Liu J, Maimaitiyiming Y, Deng Z, Li B, Su K, Chen J, Sun S, Zheng W, et al. YTHDF2-mediated regulations bifurcate BHPF-induced programmed cell deaths. Nat Sci Rev. 2023. https://doi.org/10.1093/nsr/nwad227.
Guirguis A, Ofir-Rosenfeld Y, Knezevic K, Blackaby W, Hardick D, Chan Y, Motazedian A, Gillespie A, Vassiliadis D, Lam E, et al. Inhibition of METTL3 results in a cell-intrinsic interferon response that enhances antitumor immunity. Cancer Discov. 2023;13(10):2228–47.
Wang L, Dou X, Chen S, Yu X, Huang X, Zhang L, Chen Y, Wang J, Yang K, Bugno J, et al. YTHDF2 inhibition potentiates radiotherapy antitumor efficacy. Cancer Cell. 2023;41(7):1294-1308.e1298.
Kan R, Chen J, Sallam T. Crosstalk between epitranscriptomic and epigenetic mechanisms in gene regulation. Trends Genet. 2022;38(2):182–93.
Flamand M, Tegowski M, Meyer K. The proteins of mRNA modification: writers, readers, and erasers. Annu Rev Biochem. 2023;92:145–73.
Shulman Z, Stern-Ginossar N. The RNA modification N-methyladenosine as a novel regulator of the immune system. Nat Immunol. 2020;21(5):501–12.
Deng X, Su R, Weng H, Huang H, Li Z, Chen J. RNA N-methyladenosine modification in cancers: current status and perspectives. Cell Res. 2018;28(5):507–17.
Li M, Wu X, Li G, Lv G, Wang S. FTO promotes the stemness of gastric cancer cells. DNA Cell Biol. 2023;42(7):411–20.
González-Montero J, Rojas C, Burotto M. MAP4K4 and cancer: ready for the main stage? Front Oncol. 2023;13:1162835.
Gao X, Gao C, Liu G, Hu J. MAP4K4: an emerging therapeutic target in cancer. Cell Biosci. 2016;6:56.
Singh S, Roy R, Kumar S, Srivastava P, Jha S, Rana B, Rana A. Molecular insights of MAP4K4 signaling in inflammatory and malignant diseases. Cancers. 2023. https://doi.org/10.3390/cancers15082272.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 81974385).
Author information
Authors and Affiliations
Contributions
ZY and XG designed the study. ZY and XL carried out the experiment. ZY collected and analysed data. ZY and XG wrote and revised the manuscript. ZW was responsible for supervising the study. All authors read and gave final approval of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yin, Z., Guo, X., Liang, X. et al. FTO promotes gastric cancer progression by modulating MAP4K4 expression via demethylation in an m6A-dependent manner. Med Oncol 41, 120 (2024). https://doi.org/10.1007/s12032-024-02369-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-024-02369-7