Abstract
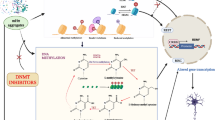
This study explores the molecular underpinnings of neuropathic pain (NPP) and neuroinflammation, focusing on the role of TRIM28 in the regulation of autophagy and microglia ferroptosis. Leveraging transcriptomic data associated with NPP, we identified TRIM28 as a critical regulator of ferroptosis. Through comprehensive analysis, including Gene Ontology enrichment and protein-protein interaction network assessments, we unveiled GSK3B as a downstream target of TRIM28. Experimental validation confirmed the capacity of TRIM28 to suppress GSK3B expression and attenuate autophagic processes in microglia. We probed the consequences of autophagy and ferroptosis on microglia physiology, iron homeostasis, oxidative stress, and the release of proinflammatory cytokines. In a murine model, we validated the pivotal role of TRIM28 in NPP and neuroinflammation. Our analysis identified 20 ferroptosis regulatory factors associated with NPP, with TRIM28 emerging as a central orchestrator. Experimental evidence affirmed that TRIM28 governs microglial iron homeostasis and cell fate by downregulating GSK3B expression and modulating autophagy. Notably, autophagy was found to influence oxidative stress and proinflammatory cytokine release through the iron metabolism pathway, ultimately fueling neuroinflammation. In vivo experiments provided conclusive evidence of TRIM28-mediated pathways contributing to heightened pain sensitivity in neuroinflammatory states. The effect of TRIM28 on autophagy and microglia ferroptosis drives NPP and neuroinflammation. These findings offer promising avenues for identifying novel therapeutic targets to manage NPP and neuroinflammation.







Similar content being viewed by others
Data Availability
The data that support the findings of this study are available on request from the corresponding author.
References
Fitzcharles MA, Cohen SP, Clauw DJ, Littlejohn G, Usui C, Häuser W (2021) Nociplastic pain: towards an understanding of prevalent pain conditions. Lancet (London England) 397(10289):2098–2110. https://doi.org/10.1016/S0140-6736(21)00392-5
Harmer B, Lee S, Duong TVH, Saadabadi A (2023) Suicidal ideation. StatPearls. StatPearls Publishing
Moloney NA, Lenoir D (2023) Assessment of neuropathic pain following cancer treatment. Anatomical record (Hoboken, N.J.: 2007), 10.1002/ar.25161. Advance online publication. https://doi.org/10.1002/ar.25161
Voute M, Morel V, Pickering G (2021) Topical Lidocaine for Chronic Pain Treatment. Drug Des Devel Ther 15:4091–4103. https://doi.org/10.2147/DDDT.S328228
Gurba KN, Chaudhry R, Haroutounian S (2022) Central Neuropathic Pain syndromes: current and emerging pharmacological strategies. CNS Drugs 36(5):483–516. https://doi.org/10.1007/s40263-022-00914-4
Berger AA, Keefe J, Winnick A, Gilbert E, Eskander JP, Yazdi C, Kaye AD, Viswanath O, Urits I (2020) Cannabis and cannabidiol (CBD) for the treatment of fibromyalgia. Best practice & research. Clin Anaesthesiol 34(3):617–631. https://doi.org/10.1016/j.bpa.2020.08.010
Jiang X, Yuan Y, Ma Y, Zhong M, Du C, Boey J, Armstrong DG, Deng W, Duan X (2021) Pain Management in people with diabetes-related chronic limb-threatening ischemia. J Diabetes Res 2021:6699292. https://doi.org/10.1155/2021/6699292
Lee JK, Hsieh IC, Su CH, Huang HL, Lei MH, Chiu KM, Huang CL, Chen CC, Hsu PC, Hsu CH, Huang CY, Lee CH, Chang WC, Lee HF, Liu JC, Yeh HI (2023) Referral, diagnosis, and Pharmacological Management of Peripheral Artery Disease: perspectives from Taiwan. Acta Cardiol Sinica 39(1):97–108. https://doi.org/10.6515/ACS.202301_39(1).20220815A
Russo MA, Baron R, Dickenson AH, Kern KU, Santarelli DM (2023) Ambroxol for neuropathic pain: hiding in plain sight? Pain 164(1):3–13. https://doi.org/10.1097/j.pain.0000000000002693
Jorfi M, Maaser-Hecker A, Tanzi RE (2023) The neuroimmune axis of Alzheimer’s disease. Genome Med 15(1):6. https://doi.org/10.1186/s13073-023-01155-w
Mert T (2023) Pulsed magnetic Field treatment for calming Neuroinflammation in Pain conditions. Altern Ther Health Med 29(6):62–67
Au NPB, Ma CHE (2022) Neuroinflammation, Microglia and implications for Retinal Ganglion Cell Survival and Axon Regeneration in Traumatic Optic Neuropathy. Front Immunol 13:860070. https://doi.org/10.3389/fimmu.2022.860070
El-Aarag SA, Mahmoud A, Hashem MH, Elkader A, Hemeida H, A. E., ElHefnawi M (2017) In silico identification of potential key regulatory factors in smoking-induced lung cancer. BMC Med Genom 10(1):40. https://doi.org/10.1186/s12920-017-0284-z
Qin Y, Li Q, Liang W, Yan R, Tong L, Jia M, Zhao C, Zhao W (2021) TRIM28 SUMOylates and stabilizes NLRP3 to facilitate inflammasome activation. Nat Commun 12(1):4794. https://doi.org/10.1038/s41467-021-25033-4
Noori T, Dehpour AR, Sureda A, Fakhri S, Sobarzo-Sanchez E, Farzaei MH, Küpeli Akkol E, Khodarahmi Z, Hosseini SZ, Alavi SD, Shirooie S (2020) The role of glycogen synthase kinase 3 beta in multiple sclerosis. Biomed Pharmacotherapy = Biomedecine Pharmacotherapie 132:110874. https://doi.org/10.1016/j.biopha.2020.110874
Kim J, Shin JY, Choi YH, Lee SY, Jin MH, Kim CD, Kang NG, Lee S (2021) Adenosine and Cordycepin Accelerate Tissue Remodeling Process through Adenosine receptor mediated Wnt/β-Catenin pathway stimulation by regulating GSK3b activity. Int J Mol Sci 22(11):5571. https://doi.org/10.3390/ijms22115571
Finnerup NB, Kuner R, Jensen TS (2021) Neuropathic Pain: from mechanisms to treatment. Physiol Rev 101(1):259–301. https://doi.org/10.1152/physrev.00045.2019
Xiao Y, Yu D (2021) Tumor microenvironment as a therapeutic target in cancer. Pharmacol Ther 221:107753. https://doi.org/10.1016/j.pharmthera.2020.107753
Ritchie ME, Phipson B, Wu D, Hu Y, Law CW, Shi W, Smyth GK (2015) Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res 43(7):e47. https://doi.org/10.1093/nar/gkv007
Tan L, Xu Q, Wang Q, Shi R, Zhang G (2020) Identification of key genes and pathways affected in epicardial adipose tissue from patients with coronary artery disease by integrated bioinformatics analysis. PeerJ 8:e8763. https://doi.org/10.7717/peerj.8763
Luo H, Ma C (2020) Identification of prognostic genes in uveal melanoma microenvironment. PLoS ONE 15(11):e0242263. https://doi.org/10.1371/journal.pone.0242263
Dai F, Wu J, Deng Z, Li H, Tan W, Yuan M, Yang D, Liu S, Zheng Y, Hu M, Yuan C, Cheng Y (2022) Integrated Bioinformatic analysis of DNA methylation and Immune Infiltration in Endometrial Cancer. Biomed Res Int 2022(5119411). https://doi.org/10.1155/2022/5119411
Shi T, Gao G (2022) Identify potential prognostic indicators and tumor-infiltrating immune cells in pancreatic adenocarcinoma. Biosci Rep 42(2):BSR20212523. https://doi.org/10.1042/BSR20212523
Yi XP, Han T, Li YX, Long XY, Li WZ (2015) Simultaneous silencing of XIAP and survivin causes partial mesenchymal-epithelial transition of human pancreatic cancer cells via the PTEN/PI3K/Akt pathway. Mol Med Rep 12(1):601–608. https://doi.org/10.3892/mmr.2015.3380
Jiang C, Tan T, Yi XP, Shen H, Li YX (2011) Lentivirus-mediated shRNA targeting XIAP and survivin inhibit SW1990 pancreatic cancer cell proliferation in vitro and in vivo. Mol Med Rep 4(4):667–674. https://doi.org/10.3892/mmr.2011.472
Kannan M, Sil S, Oladapo A, Thangaraj A, Periyasamy P, Buch S (2023) HIV-1 Tat-mediated microglial ferroptosis involves the mir-204-ACSL4 signaling axis. Redox Biol 62:102689. https://doi.org/10.1016/j.redox.2023.102689
Lin JQ, Tian H, Zhao XG, Lin S, Li DY, Liu YY, Xu C, Mei XF (2021) Zinc provides neuroprotection by regulating NLRP3 inflammasome through autophagy and ubiquitination in a spinal contusion injury model. CNS Neurosci Ther 27(4):413–425. https://doi.org/10.1111/cns.13460
Zeng Z, Li Y, Pan Y, Lan X, Song F, Sun J, Zhou K, Liu X, Ren X, Wang F, Hu J, Zhu X, Yang W, Liao W, Li G, Ding Y, Liang L (2018) Cancer-derived exosomal mir-25-3p promotes pre-metastatic niche formation by inducing vascular permeability and angiogenesis. Nat Commun 9(1):5395. https://doi.org/10.1038/s41467-018-07810-w
Ma, X. H., Piao, S. F., Dey, S., McAfee, Q., Karakousis, G., Villanueva, J., Hart, L. S., Levi, S., Hu, J., Zhang, G., Lazova, R., Klump, V., Pawelek, J. M., Xu, X., Xu, W., Schuchter, L. M., Davies, M. A., Herlyn, M., Winkler, J., Koumenis, C., … Amaravadi, R. K. (2014). Targeting ER stress-induced autophagy overcomes BRAF inhibitor resistance in melanoma. The Journal of clinical investigation, 124(3):1406–1417. https://doi.org/10.1172/JCI70454
Liao S, Wu J, Liu R, Wang S, Luo J, Yang Y, Qin Y, Li T, Zheng X, Song J, Zhao X, Xiao C, Zhang Y, Bian L, Jia P, Bai Y, Zheng X (2020) A novel compound DBZ ameliorates neuroinflammation in LPS-stimulated microglia and ischemic stroke rats: role of akt(Ser473)/GSK3β(Ser9)-mediated Nrf2 activation. Redox Biol 36:101644. https://doi.org/10.1016/j.redox.2020.101644
Park E, Chung SW (2019) ROS-mediated autophagy increases intracellular iron levels and ferroptosis by ferritin and transferrin receptor regulation. Cell Death Dis 10(11):822. https://doi.org/10.1038/s41419-019-2064-5
Zhu K, Zhu X, Sun S, Yang W, Liu S, Tang Z, Zhang R, Li J, Shen T, Hei M (2021) Inhibition of TLR4 prevents hippocampal hypoxic-ischemic injury by regulating ferroptosis in neonatal rats. Exp Neurol 345:113828. https://doi.org/10.1016/j.expneurol.2021.113828
Lu Y, Zhang J, Zeng F, Wang P, Guo X, Wang H, Qin Z, Tao T (2022) Human PMSCs-derived small extracellular vesicles alleviate neuropathic pain through miR-26a-5p/Wnt5a in SNI mice model. J Neuroinflamm 19(1):221. https://doi.org/10.1186/s12974-022-02578-9
Zeng H, Liu N, Yang YY, Xing HY, Liu XX, Li F, La GY, Huang MJ, Zhou MW (2019) Lentivirus-mediated downregulation of α-synuclein reduces neuroinflammation and promotes functional recovery in rats with spinal cord injury. J Neuroinflamm 16(1):283. https://doi.org/10.1186/s12974-019-1658-2
Li YJ, Chen X, Kwan TK, Loh YW, Singer J, Liu Y, Ma J, Tan J, Macia L, Mackay CR, Chadban SJ, Wu H (2020) Dietary Fiber protects against Diabetic Nephropathy through short-chain fatty acid-mediated activation of G protein-coupled receptors GPR43 and GPR109A. J Am Soc Nephrology: JASN 31(6):1267–1281. https://doi.org/10.1681/ASN.2019101029
Wu C, Pan Y, Wang L, Liu M, Wu M, Wang J, Yang G, Guo Y, Ma Y (2022) A new method for primary culture of microglia in rats with spinal cord injury. Biochem Biophys Res Commun 599:63–68. https://doi.org/10.1016/j.bbrc.2022.02.027
Deng YF, Xiang P, Du JY, Liang JF, Li X (2023) Intrathecal liproxstatin-1 delivery inhibits ferroptosis and attenuates mechanical and thermal hypersensitivities in rats with complete Freund’s adjuvant-induced inflammatory pain. Neural Regeneration Res 18(2):456–462. https://doi.org/10.4103/1673-5374.346547
Qian ZY, Kong RY, Zhang S, Wang BY, Chang J, Cao J, Wu CQ, Huang ZY, Duan A, Li HJ, Yang L, Cao XJ (2022) Ruxolitinib attenuates secondary injury after traumatic spinal cord injury. Neural Regeneration Res 17(9):2029–2035. https://doi.org/10.4103/1673-5374.335165
Hosseini M, Yousefifard M, Aziznejad H, Nasirinezhad F, Transplantation (2015) 21(9), 1537–1544. https://doi.org/10.1016/j.bbmt.2015.05.008
Gao X, Gao LF, Kong XQ, Zhang YN, Jia S, Meng CY (2023) Mesenchymal stem cell-derived extracellular vesicles carrying miR-99b-3p restrain microglial activation and neuropathic pain by stimulating autophagy. Int Immunopharmacol 115:109695. https://doi.org/10.1016/j.intimp.2023.109695
Czerwińska P, Mazurek S, Wiznerowicz M (2017) The complexity of TRIM28 contribution to cancer. J Biomed Sci 24(1):63. https://doi.org/10.1186/s12929-017-0374-4
Peng Y, Zhang M, Jiang Z, Jiang Y (2019) TRIM28 activates autophagy and promotes cell proliferation in glioblastoma. OncoTargets Therapy 12:397–404. https://doi.org/10.2147/OTT.S188101
Song T, Lv S, Ma X, Zhao X, Fan L, Zou Q, Li N, Yan Y, Zhang W, Sun L (2023) TRIM28 represses renal cell carcinoma cell proliferation by inhibiting TFE3/KDM6A-regulated autophagy. J Biol Chem 299(5):104621. https://doi.org/10.1016/j.jbc.2023.104621
Pineda CT, Potts PR (2015) Oncogenic MAGEA-TRIM28 ubiquitin ligase downregulates autophagy by ubiquitinating and degrading AMPK in cancer. Autophagy 11(5):844–846. https://doi.org/10.1080/15548627.2015.1034420
Zhou B, Liu J, Kang R, Klionsky DJ, Kroemer G, Tang D (2020) Ferroptosis is a type of autophagy-dependent cell death. Sem Cancer Biol 66:89–100. https://doi.org/10.1016/j.semcancer.2019.03.002
Ryu HY, Kim LE, Jeong H, Yeo BK, Lee JW, Nam H, Ha S, An HK, Park H, Jung S, Chung KM, Kim J, Lee BH, Cheong H, Kim EK, Yu SW (2021) GSK3B induces autophagy by phosphorylating ULK1. Exp Mol Med 53(3):369–383. https://doi.org/10.1038/s12276-021-00570-6
Chen J, Qin C, Zhou Y, Chen Y, Mao M, Yang J (2022) Metformin may induce ferroptosis by inhibiting autophagy via lncRNA H19 in breast cancer. FEBS open bio 12(1):146–153. https://doi.org/10.1002/2211-5463.13314
Sung HK, Murugathasan M, Abdul-Sater AA, Sweeney G (2023) Autophagy deficiency exacerbates iron overload induced reactive oxygen species production and apoptotic cell death in skeletal muscle cells. Cell Death Dis 14(4):252. https://doi.org/10.1038/s41419-022-05484-3
Xie Y, Hou W, Song X, Yu Y, Huang J, Sun X, Kang R, Tang D (2016) Ferroptosis: process and function. Cell Death Differ 23(3):369–379. https://doi.org/10.1038/cdd.2015.158
Mauvezin C, Neufeld TP (2015) Bafilomycin A1 disrupts autophagic flux by inhibiting both V-ATPase-dependent acidification and Ca-P60A/SERCA-dependent autophagosome-lysosome fusion. Autophagy 11(8):1437–1438. https://doi.org/10.1080/15548627.2015.1066957
Theil EC (2004) Iron, ferritin, and nutrition. Annu Rev Nutr 24:327–343. https://doi.org/10.1146/annurev.nutr.24.012003.132212
Liu S, Gao X, Zhou S (2022) New Target for Prevention and Treatment of Neuroinflammation: Microglia Iron Accumulation and Ferroptosis. ASN neuro, 14, 17590914221133236. https://doi.org/10.1177/17590914221133236
Liang D, Minikes AM, Jiang X (2022) Ferroptosis at the intersection of lipid metabolism and cellular signaling. Mol Cell 82(12):2215–2227. https://doi.org/10.1016/j.molcel.2022.03.022
Klionsky, D. J., Petroni, G., Amaravadi, R. K., Baehrecke, E. H., Ballabio, A., Boya, P., Bravo-San Pedro, J. M., Cadwell, K., Cecconi, F., Choi, A. M. K., Choi, M. E., Chu, C. T., Codogno, P., Colombo, M. I., Cuervo, A. M., Deretic, V., Dikic, I., Elazar, Z., Eskelinen, E. L., Fimia, G. M., … Pietrocola, F. (2021). Autophagy in major human diseases. The EMBO journal, 40(19):e108863. https://doi.org/10.15252/embj.2021108863
Li J, Tian M, Hua T, Wang H, Yang M, Li W, Zhang X, Yuan H (2021) Combination of autophagy and NFE2L2/NRF2 activation as a treatment approach for neuropathic pain. Autophagy 17(12):4062–4082. https://doi.org/10.1080/15548627.2021.1900498
Bolisetty S, Zarjou A, Agarwal A (2017) Heme Oxygenase 1 as a therapeutic target in Acute kidney Injury. Am J Kidney Diseases: Official J Natl Kidney Foundation 69(4):531–545. https://doi.org/10.1053/j.ajkd.2016.10.037
Wang C, Zhang R, Wei X, Lv M, Jiang Z (2020) Metalloimmunology: the metal ion-controlled immunity. Adv Immunol 145:187–241. https://doi.org/10.1016/bs.ai.2019.11.007
Cui X, Chen F, Zhao J, Li D, Hu M, Chen X, Zhang Y, Han L (2023) Involvement of JNK signaling in Aspergillus fumigatus-induced inflammatory factors release in bronchial epithelial cells. Sci Rep 13(1):1293. https://doi.org/10.1038/s41598-023-28567-3
Wang K, Li Z, Li Y, Liu X, Sun Y, Hong J, Ding Y, Zheng W, Qian L, Xu D (2022) Cardioprotection of Klotho against myocardial infarction-induced heart failure through inducing autophagy. Mech Ageing Dev 207:111714. https://doi.org/10.1016/j.mad.2022.111714
Xiao X, Liu D, Chen S, Li X, Ge M, Huang W (2021) Sevoflurane preconditioning activates HGF/Met-mediated autophagy to attenuate hepatic ischemia-reperfusion injury in mice. Cell Signal 82:109966. https://doi.org/10.1016/j.cellsig.2021.109966
Liang, J., Wang, L., Wang, C., Shen, J., Su, B., Marisetty, A. L., Fang, D., Kassab, C., Jeong, K. J., Zhao, W., Lu, Y., Jain, A. K., Zhou, Z., Liang, H., Sun, S. C., Lu, C., Xu, Z. X., Yu, Q., Shao, S., Chen, X., … Heimberger, A. B. (2020). Verteporfin Inhibits PD-L1 through Autophagy and the STAT1-IRF1-TRIM28 Signaling Axis, Exerting Antitumor Efficacy. Cancer immunology research, 8(7):952–965. https://doi.org/10.1158/2326-6066.CIR-19-0159
Xu W, Li J, He C, Wen J, Ma H, Rong B, Diao J, Wang L, Wang J, Wu F, Tan L, Shi YG, Shi Y, Shen H (2021) METTL3 regulates heterochromatin in mouse embryonic stem cells. Nature 591(7849):317–321. https://doi.org/10.1038/s41586-021-03210-1
Tovo PA, Davico C, Marcotulli D, Vitiello B, Daprà V, Calvi C, Montanari P, Carpino A, Galliano I, Bergallo M (2022) Enhanced expression of human endogenous retroviruses, TRIM28 and SETDB1 in Autism Spectrum Disorder. Int J Mol Sci 23(11):5964. https://doi.org/10.3390/ijms23115964
Chen X, Zhao Y (2019) Genetic involvement in Dental Implant failure: Association with polymorphisms of genes modulating inflammatory responses and bone metabolism. J Oral Implantol 45(4):318–326. https://doi.org/10.1563/aaid-joi-D-18-00212
Goldstein N, McKnight AD, Carty JRE, Arnold M, Betley JN, Alhadeff AL (2021) Hypothalamic detection of macronutrients via multiple gut-brain pathways. Cell Metabol 33(3):676–687e5. https://doi.org/10.1016/j.cmet.2020.12.018
Liu M, Luo G, Dong L, Mazhar M, Wang L, He W, Liu Y, Wu Q, Zhou H, Yang S (2022) Network Pharmacology and in Vitro Experimental Verification reveal the mechanism of the Hirudin in suppressing myocardial hypertrophy. Front Pharmacol 13:914518. https://doi.org/10.3389/fphar.2022.914518
Darbari DS, Sheehan VA, Ballas SK (2020) The vaso-occlusive pain crisis in sickle cell disease: definition, pathophysiology, and management. Eur J Haematol 105(3):237–246. https://doi.org/10.1111/ejh.13430
Cole JB, Florez JC (2020) Genetics of diabetes mellitus and diabetes complications. Nat Rev Nephrol 16(7):377–390. https://doi.org/10.1038/s41581-020-0278-5
Jiang Z, Guo N, Hong K (2020) A three-tiered integrative analysis of transcriptional data reveals the shared pathways related to heart failure from different aetiologies. J Cell Mol Med 24(16):9085–9096. https://doi.org/10.1111/jcmm.15544
Li L, Li T, Qu X, Sun G, Fu Q, Han G (2023) Stress/cell death pathways, neuroinflammation, and neuropathic pain. Immunol Rev. https://doi.org/10.1111/imr.13275Advance online publication
Liao MF, Lu KT, Hsu JL, Lee CH, Cheng MY, Ro LS (2022) The role of Autophagy and apoptosis in Neuropathic Pain formation. Int J Mol Sci 23(5):2685. https://doi.org/10.3390/ijms23052685
Li L, Guo L, Gao R, Yao M, Qu X, Sun G, Fu Q, Hu C, Han G (2023) Ferroptosis: a new regulatory mechanism in neuropathic pain. Front Aging Neurosci 15:1206851. https://doi.org/10.3389/fnagi.2023.1206851
Liu J, Kuang F, Kroemer G, Klionsky DJ, Kang R, Tang D (2020) Autophagy-dependent ferroptosis: Machinery and Regulation. Cell Chem Biology 27(4):420–435. https://doi.org/10.1016/j.chembiol.2020.02.005
Mancias JD, Wang X, Gygi SP, Harper JW, Kimmelman AC (2014) Quantitative proteomics identifies NCOA4 as the cargo receptor mediating ferritinophagy. Nature 509(7498):105–109. https://doi.org/10.1038/nature13148
Chen X, Li J, Kang R, Klionsky DJ, Tang D (2021) Ferroptosis: machinery and regulation. Autophagy 17(9):2054–2081. https://doi.org/10.1080/15548627.2020.1810918
Lei K, Wu R, Wang J, Lei X, Zhou E, Fan R, Gong L (2023) Sirtuins as potential targets for Neuroprotection: mechanisms of early brain Injury Induced by Subarachnoid Hemorrhage. Translational stroke research, https://doi.org/10.1007/s12975-023-01191-z. https://doi.org/10.1007/s12975-023-01191-z. Advance online publication
Han J, Wang Y, Zhou H, Ai S, Wan D (2022) Integrated Bioinformatics and experimental analysis identified TRIM28 a potential prognostic biomarker and correlated with Immune infiltrates in Liver Hepatocellular Carcinoma. Comput Math Methods Med 2022(6267851). https://doi.org/10.1155/2022/6267851
Wu L, Yu D, Liu P, Gao C, Wang Z (2023) Heuristic heterogeneous graph reasoning networks for Fact Verification. IEEE Trans Neural Networks Learn Syst PP. https://doi.org/10.1109/TNNLS.2023.3282380. https://doi.org/10.1109/TNNLS.2023.3282380 Advance online publication
Acknowledgements
None.
Funding
This study was supported by The Sichuan Science and Technology Program (2023YFS0254), Central Nervous System Drug Key Laboratory of Sichuan Province, Southwest Medical University (230006-01SZ), Sichuan Science and Technology Program (2021YJ0181), and Southwest Medical University School Program (2023QN005).
Author information
Authors and Affiliations
Contributions
JT, QC and LX wrote the paper and conceived and designed the experiments; TT and YZ analyzed the data; CHO collected and provided the sample for this study. All authors have read and approved the final submitted manuscript.
Corresponding authors
Ethics declarations
Ethics Approval
The Experimental Procedures and Animal Use Plans for this study were approved by the Animal Ethics Committee of the Affiliated Traditional Chinese Medicine Hospital (NO. 20220815- 004).
Consent to Participate
Not applicable.
Consent for publication
Consent for publication was obtained from the participants.
Competing interests
The author declares no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tang, J., Chen, Q., Xiang, L. et al. TRIM28 Fosters Microglia Ferroptosis via Autophagy Modulation to Enhance Neuropathic Pain and Neuroinflammation. Mol Neurobiol (2024). https://doi.org/10.1007/s12035-024-04133-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12035-024-04133-4