Abstract
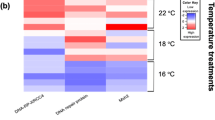
Thermal variations due to global climate change are expected to modify the distributions of marine ectotherms, with potential pathogen translocations. This is of particular concern at high latitudes where cold-adapted stenothermal fish such as the Notothenioids occur. However, little is known about the combined effects of thermal fluctuations and immune challenges on the balance between cell damage and repair processes in these fish. The aim of this study was to determine the effect of thermal variation on specific genes involved in the ubiquitination and apoptosis pathways in two congeneric Notothenioid species, subjected to simulated bacterial and viral infections. Adult fish of Harpagifer bispinis and Harpagifer antarcticus were collected from Punta Arenas (Chile) and King George Island (Antarctica), respectively, and distributed as follows: injected with PBS (control), LPS (2.5 mg/kg) or Poly I:C (2 mg/kg) and then submitted to 2, 5 and 8 °C. After 1 week, samples of gills, liver and spleen were taken to evaluate the expression by real-time PCR of specific genes involved in ubiquitination (E3-ligase enzyme) and apoptosis (BAX and SMAC/DIABLO). Gene expression was tissue-dependent and increased with increasing temperature in the gills and liver while showing an opposite pattern in the spleen. Studying a pair of sister species that occur across the Antarctic Polar Front can help us understand the particular pressures of intertidal lifestyles and the effect of temperature in combination with biological stressors on cell damage and repair capacity in a changing environment.



Similar content being viewed by others
Data availability
All data is available in this dataset link: https://doi.org/https://doi.org/10.5281/zenodo.8029858.
References
Ahn DH, Kang S, Park H (2016) Transcriptome analysis of immune response genes induced by pathogen agonists in the Antarctic bullhead notothen Notothenia coriiceps. Fish Shellfish Immunol 55:315–322. https://doi.org/10.1016/j.fsi.2016.06.004
Ametrano A, Picchietti S, Guerra L et al (2022) Comparative analysis of the pIgR gene from the Antarctic Teleost Trematomus bernacchii reveals distinctive features of cold-adapted Notothenioidei. Int J Mol Sci 23(14):7783. https://doi.org/10.3390/IJMS23147783
Angilletta MJ (2009) Thermal adaptation: a theoretical and empirical synthesis. Oxford University Press
Avendaño-Herrera R, Irgang R, Poblete-Morales M et al (2016) Predominant bacteria associated with black rockcod (Notothenia coriiceps, Richardson 1844) from King George Island. Antarctica 36:246–255
Ben-Shachar M, Lüdecke D, Makowski D (2020) effectsize: estimation of effect size indices and standardized parameters. J Open Source Softw 5:2815. https://doi.org/10.21105/JOSS.02815
Bilyk KT, Cheng CHC (2014) RNA-seq analyses of cellular responses to elevated body temperature in the high Antarctic cryopelagic nototheniid fish Pagothenia borchgrevinki. Mar Genomics 18:163–171. https://doi.org/10.1016/j.margen.2014.06.006
Borley KA, Beers JM, Sidell BD (2010) Phenylhydrazine-induced anemia causes nitric-oxide-mediated upregulation of the angiogenic pathway in Notothenia coriiceps. J Exp Biol 213:2865–2872. https://doi.org/10.1242/jeb.043281
Cárdenas L, Leclerc J-C, Bruning P et al (2020) First mussel settlement observed in Antarctica reveals the potential for future invasions. Sci Rep 10:5552. https://doi.org/10.1038/s41598-020-62340-0
Carney Almroth B, Asker N, Wassmur B et al (2015) Warmer water temperature results in oxidative damage in an Antarctic fish, the bald notothen. J Exp Mar Bio Ecol 468:130–137. https://doi.org/10.1016/j.jembe.2015.02.018
Cheng CH, Yang FF, Liao SA et al (2015) High temperature induces apoptosis and oxidative stress in pufferfish (Takifugu obscurus) blood cells. J Therm Biol 53:172–179. https://doi.org/10.1016/j.jtherbio.2015.08.002
Ciechanover A (1998) The ubiquitin-proteasome pathway: on protein death and cell life. EMBO J 17:7151–7160. https://doi.org/10.1093/emboj/17.24.7151
Clark MS, Fraser KPP, Burns G, Peck LS (2008) The HSP70 heat shock response in the Antarctic fish Harpagifer antarcticus. Polar Biol 31:171–180. https://doi.org/10.1007/s00300-007-0344-5
Clarke A, Murphy EJ, Meredith MP et al (2007) Climate change and the marine ecosystem of the western Antarctic Peninsula. Philos Trans R Soc B Biol Sci 362:149–166. https://doi.org/10.1098/rstb.2006.1958
Comte L, Olden JD (2017) Climatic vulnerability of the world’s freshwater and marine fishes. Nat Clim Chang 7:718–722. https://doi.org/10.1038/nclimate3382
Cowan DA, Chown SL, Convey P et al (2011) Non-indigenous microorganisms in the Antarctic: assessing the risks. Trends Microbiol 19:540–548. https://doi.org/10.1016/J.TIM.2011.07.008
Crain CM, Kroeker K, Halpern BS (2008) Interactive and cumulative effects of multiple human stressors in marine systems. Ecol Lett 11:1304–1315. https://doi.org/10.1111/J.1461-0248.2008.01253.X
Davis ME, Gack MU (2015) Ubiquitination in the antiviral immune response. Virology 479–480:52–65. https://doi.org/10.1016/J.VIROL.2015.02.033
Eastman JT (2005) The nature of the diversity of Antarctic fishes. Polar Biol 28:93–107. https://doi.org/10.1007/s00300-004-0667-4
Eastman JT, Eakin RR (2000) An updated species list for notothenioid fish (Perciformes; Notothenioidei ), with comments on Antarctic species. Arch Fish Mar Res 48:11–20
Fänge R, Nilsson S (1985) The fish spleen: structure and function. Experientia 41:152–158. https://doi.org/10.1007/BF02002607
Fulda S, Gorman AM, Hori O, Samali A (2010) Cellular stress responses: cell survival and cell death. Int J Cell Biol 2010:1–23. https://doi.org/10.1155/2010/214074
Giménez EM, Barrantes ME, Fernández DA, Lattuca ME (2021) Thermal responses of two sub-Antarctic notothenioid fishes, the black southern cod Patagonotothen tessellata (Richardson, 1845) and the Magellan plunderfish Harpagifer bispinis (Forster, 1801), from southern South America. Polar Biol 44:1055–1067. https://doi.org/10.1007/s00300-021-02852-1
Guijarro JA, Cascales D, García-Torrico AI et al (2015) Temperature-dependent expression of virulence genes in fish-pathogenic bacteria. Front Microbiol 6:700. https://doi.org/10.3389/fmicb.2015.00700
Harvell CD, Kim K, Burkholder JM et al (1999) Emerging marine diseases–climate links and anthropogenic factors. Science 285(5433):1505–1510. https://doi.org/10.1126/science.285.5433.1505
Hüne M, González-Wevar C, Poulin E et al (2015) Low level of genetic divergence between Harpagifer fish species (Perciformes: Notothenioidei) suggests a quaternary colonization of Patagonia from the Antarctic Peninsula. Polar Biol 38:607–617. https://doi.org/10.1007/s00300-014-1623-6
Hureau JC (1990) Harpagiferidae. Fishes south ocean. In: Gon O, Heemstra PC, Smith JLB (eds) Inst Ichthyol Grahamst, South Africa. pp 357–363
Kiraz Y, Adan A, Kartal Yandim M, Baran Y (2016) Major apoptotic mechanisms and genes involved in apoptosis. Tumor Biol 37:8471–8486. https://doi.org/10.1007/s13277-016-5035-9
Kroemer G, Galluzzi L, Brenner C (2007) Mitochondrial membrane permeabilization in cell death. Physiol Rev 87:99–163. https://doi.org/10.1152/physrev.00013.2006
La Mesa M, Vera-Duarte J, Landaeta MF (2017) Early life history traits of Harpagifer antarcticus (Harpagiferidae, Notothenioidei) from the South Shetland Islands during austral summer. Polar Biol 40:1699–1705. https://doi.org/10.1007/s00300-017-2093-4
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Logan CA, Somero GN (2011) Effects of thermal acclimation on transcriptional responses to acute heat stress in the eurythermal fish Gillichthys mirabilis (Cooper). Am J Physiol Regul Integr Comp Physiol 300:R1373–R1383. https://doi.org/10.1152/ajpregu.00689.2010
Makrinos DL, Bowden TJ (2016) Natural environmental impacts on teleost immune function. Fish Shellfish Immunol 53:50–57. https://doi.org/10.1016/J.FSI.2016.03.008
Martínez D, Vargas-Lagos C, Saravia J et al (2020) Cellular stress responses of Eleginops maclovinus fish injected with Piscirickettsia salmonis and submitted to thermal stress. Cell Stress Chaperones 25:93–104. https://doi.org/10.1007/s12192-019-01051-6
Martínez D, Moncada-Kopp C, Paschke K et al (2021) Warming and freshening activate the transcription of genes involved in the cellular stress response in Harpagifer antarcticus. Fish Physiol Biochem 47:533–546. https://doi.org/10.1007/s10695-021-00931-y
McCarthy AH, Peck LS, Hughes KA, Aldridge DC (2019) Antarctica: the final frontier for marine biological invasions. Glob Chang Biol 25:2221–2241. https://doi.org/10.1111/GCB.14600
McKenna S, García-Gutiérrez L, Matallanas D, Fey D (2021) BAX and SMAC regulate bistable properties of the apoptotic caspase system. Sci Rep 11:1–15. https://doi.org/10.1038/s41598-021-82215-2
Meredith MP, King JC (2005) Rapid climate change in the ocean west of the Antarctic Peninsula during the second half of the 20th century. Geophys Res Lett 32:1–5. https://doi.org/10.1029/2005GL024042
Navarro JM, Paschke K, Ortiz A et al (2019) The Antarctic fish Harpagifer antarcticus under current temperatures and salinities and future scenarios of climate change. Prog Oceanogr 174:37–43. https://doi.org/10.1016/j.pocean.2018.09.001
Pachauri RK, Meyer LA (2014) Climate change 2014: synthesis report. Contribution of working groups I, II and III to the fifth assessment report of the intergovernmental panel on climate change. Geneva, Switzerland, Cambridge University Press
Philip AM, Jørgensen EH, Maule AG, Vijayan MM (2018) Extended fasting does not affect the liver innate immune response in rainbow trout. Dev Comp Immunol 79:67–74. https://doi.org/10.1016/j.dci.2017.10.009
Pontigo JP, Saravia J, Oyarzún R et al (2019) Modulation of the expression of immune-related gene in atlantic and coho salmon during infestation with the sea lice caligus rogercresseyi. Fishes 4:42. https://doi.org/10.3390/fishes4030042
Pörtner HO, Farrell AP (2008) Ecology: physiology and climate change. Science 322(5902):690–692. https://doi.org/10.1126/science.1163156
Rasmussen L (2004) Rapid cycle real-time PCR — methods and applications, rapid cycle real-time PCR — methods and applications. Springer Berlin Heidelberg. https://doi.org/10.1007/978-3-642-18840-4
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing
Richter K, Haslbeck M, Buchner J (2010) The heat shock response: life on the verge of death. Mol Cell 40:253–266. https://doi.org/10.1016/j.molcel.2010.10.006
Saravia J, Paschke K, Oyarzún-Salazar R et al (2021) Effects of warming rates on physiological and molecular components of response to CTMax heat stress in the Antarctic fish Harpagifer antarcticus. J Therm Biol 99:103021. https://doi.org/10.1016/j.jtherbio.2021.103021
Saravia J, Paschke K, Pontigo JP et al (2022) Effects of temperature on the innate immune response on Antarctic and sub-Antarctic fish Harpagifer antarcticus and Harpagifer bispinis challenged with two immunostimulants, LPS and Poly I:C: In vivo and in vitro approach. Fish Shellfish Immunol 130:391–408. https://doi.org/10.1016/J.FSI.2022.09.025
Schreck CB (2016) The concept of stress in fish. Elsevier Inc, First Edit
Segovia NI, González-Wevar CA, Naretto J, et al (2022) The right tool for the right question: contrasting biogeographic patterns in the notothenioid fish Harpagifer spp. along the Magellan Province. Proc R Soc B Biol Sci 289:20212738. https://doi.org/10.1098/rspb.2021.2738
Sherman MY, Goldberg AL (2001) Cellular defenses against unfolded proteins. Neuron 29:15–32. https://doi.org/10.1016/S0896-6273(01)00177-5
Sleadd IM, Lee M, Hassumani DO et al (2014) Sub-lethal heat stress causes apoptosis in an Antarctic fish that lacks an inducible heat shock response. J Therm Biol 44:119–125. https://doi.org/10.1016/j.jtherbio.2014.06.007
Somero GN (2010) The physiology of climate change: how potentials for acclimatization and genetic adaptation will determine ‘winners’ and ‘losers.’ J Exp Biol 213:912–920
Somero GN (2020) The cellular stress response and temperature: function, regulation, and evolution. J Exp Zool Part A Ecol Integr Physiol 333:379–397. https://doi.org/10.1002/JEZ.2344
Todgham AE, Crombie TA, Hofmann GE (2017) The effect of temperature adaptation on the ubiquitin–proteasome pathway in notothenioid fishes. J Exp Biol 220:369–378. https://doi.org/10.1242/jeb.145946
Turner J, Colwell SR, Marshall GJ et al (2005) Antarctic climate change during the last 50 years. Int J Climatol 25:279–294. https://doi.org/10.1002/joc.1130
Vargas-Chacoff L, Muñoz JLP, Hawes C et al (2016) Atlantic salmon (Salmo salar) and Coho salmon (Oncorhynchus kisutch) display differential metabolic changes in response to infestation by the ectoparasite Caligus rogercresseyi. Aquaculture 464:469–479. https://doi.org/10.1016/j.aquaculture.2016.07.029
Vargas-Chacoff L, Arjona FJ, Ruiz-Jarabo I et al (2020) Water temperature affects osmoregulatory responses in gilthead sea bream (Sparus aurata L.). J Therm Biol 88:102526. https://doi.org/10.1016/j.jtherbio.2020.102526
Vargas-Chacoff L, Dann F, Paschke K et al (2021a) Freshening effect on the osmotic response of the Antarctic spiny plunderfish Harpagifer antarcticus. J Fish Biol 98:1558–1571. https://doi.org/10.1111/JFB.14676
Vargas-Chacoff L, Martínez D, Oyarzún-Salazar R et al (2021b) The osmotic response capacity of the Antarctic fish Harpagifer antarcticus is insufficient to cope with projected temperature and salinity under climate change. J Therm Biol 96:102835. https://doi.org/10.1016/j.jtherbio.2021.102835
Wendelaar Bonga SE (1997) The stress response in fish. Physiol Rev 77:591–625. https://doi.org/10.1152/physrev.1997.77.3.591
White MJ (1992) Reproduction and larval growth of Harpagifer antarcticus Nybelin (Pisces, Notothenioidei). Antarct Sci 4:421–430. https://doi.org/10.1017/S0954102092000622
Windisch HS, Frickenhaus S, John U et al (2014) Stress response or beneficial temperature acclimation: Transcriptomic signatures in Antarctic fish (Pachycara brachycephalum). Mol Ecol 23:3469–3482. https://doi.org/10.1111/mec.12822
Wong BBM, Candolin U (2015) Behavioral responses to changing environments. Behav Ecol 26:665–673. https://doi.org/10.1093/BEHECO/ARU183
Yang Q, Zhao J, Chen D (2021) Wang Y (2021) E3 ubiquitin ligases: styles, structures and functions. Mol Biomed 21(2):1–17. https://doi.org/10.1186/S43556-021-00043-2
Zhou ZX, Zhang BC, Sun L (2014) Poly(I:C) induces antiviral immune responses in Japanese flounder (Paralichthys olivaceus) that require TLR3 and MDA5 and is negatively regulated by Myd88. PLoS ONE 9:e112918. https://doi.org/10.1371/journal.pone.0112918
Zhou Z, He Y, Wang S et al (2022) Autophagy regulation in teleost fish: a double-edged sword. Aquaculture 558:738369
Zinngrebe J, Montinaro A, Peltzer N, Walczak H (2014) Ubiquitin in the immune system. EMBO Rep 15:28–45. https://doi.org/10.1002/EMBR.201338025
Acknowledgements
The authors also wish to acknowledge Instituto Antártico Chileno (INACH) for its support in Antarctica and appreciate the collaboration between CONICYT (now ANID) and INACH in the improvement of the “Professor Julio Escudero” base, which has favoured the fieldwork. The authors also want to thank the anonymous reviewers and L Verde Arregoitia for their comments that helped greatly to improve this manuscript. The authors acknowledge Vicerrectoría de Investigación (VIDCA) and Programa de Doctorado en Ciencias de la Acuicultura (Universidad Austral de Chile) for their support.
Funding
This work was supported by Fondap-Ideal Grant No. 15150003, ANID-Millennium Science Initiative Program-Center ICM-ANID ICN2021_002 and Instituto Antártico Chileno (INACH) grant number DG_13-20. JS acknowledges Agencia Nacional de Investigación y Desarrollo (CONICyT now ANID, Folio 21170636) and is presently funded by ANID Fondecyt de Postdoctorado (Folio 3230234).
Author information
Authors and Affiliations
Contributions
L. Vargas-Chacoff, K. Paschke and J. M. Navarro designed the experiment, and they were in Antarctica. J. Saravia, J. P. Pontigo, K. Paschke, J. M. Navarro and L. Vargas-Chacoff did the sampling. J. Saravia, J. P. Pontigo and D. Nualart analysed the samples. J. Saravia and L. Vargas-Chacoff did the graph and statistical analysis. J. Saravia, J. P. Pontigo, D. Nualart, K. Paschke, J. M. Navarro and L. Vargas-Chacoff revised the draft. J. Saravia, J. P. Pontigo, D. Nualart, K. Paschke, J. M. Navarro and L. Vargas-Chacoff revised the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All procedures were performed following the guidelines regulating the use of laboratory animals established by Universidad Austral de Chile (UACh) and Agencia Nacional de Investigación y Desarrollo (ANID).
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saravia, J., Nualart, D., Paschke, K. et al. Temperature and immune challenges modulate the transcription of genes of the ubiquitin and apoptosis pathways in two high-latitude Notothenioid fish across the Antarctic Polar Front. Fish Physiol Biochem (2024). https://doi.org/10.1007/s10695-024-01348-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10695-024-01348-z